Chapter 19: Q. 31 (page 544)
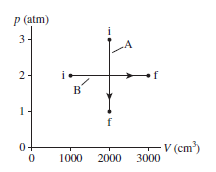
0.10 mol of nitrogen gas follow the two processes shown in
Figure EX19.31. How much heat is required for each?
Short Answer
The heat required for different process are -1000 j and 700 J.
Learning Materials
Features
Discover
Chapter 19: Q. 31 (page 544)
0.10 mol of nitrogen gas follow the two processes shown in
Figure EX19.31. How much heat is required for each?

The heat required for different process are -1000 j and 700 J.
All the tools & learning materials you need for study success - in one app.
Get started for free
Why is the molar specific heat of a gas at constant pressure larger than the molar specific heat at constant volume?
a. 100 J of heat energy are transferred to 20 g of mercury. By how much does the temperature increase?
b. How much heat is needed to raise the temperature of 20 g of water by the same amount?
of nitrogen gas at and an initial pressure of undergo an isobaric expansion until the volume has tripled.
a. What are the gas volume and temperature after the expansion?
b. How much heat energy is transferred to the gas to cause this expansion?
The gas pressure is then decreased at constant volume until the original temperature is reached.
c. What is the gas pressure after the decrease?
d. What amount of heat energy is transferred from the gas as its pressure decreases?
e. Show the total process on a diagram. Provide an appropriate scale on both axes.
When a space capsule returns to earth, its surfaces get very hot as it passes through the atmosphere at high speed. Has the space capsule been heated? If so, what was the source of the heat? If not, why is it hot?
a. Is the final temperature higher than, lower than, or equal to the initial temperature?
b. Is any heat energy added to or removed from the system in this process? Explain
What do you think about this solution?
We value your feedback to improve our textbook solutions.