Chapter 19: Q. 7 (page 543)
w
Learning Materials
Features
Discover
Chapter 19: Q. 7 (page 543)
w
All the tools & learning materials you need for study success - in one app.
Get started for free
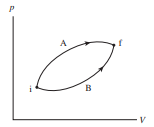
Shows two different processes taking an ideal gas from state i to state f.
a. Is the temperature change ∆T during process A larger than, smaller than, or equal to the change during process B? Explain.
b. Is the heat energy added during process A greater than, less than, or equal to the heat added during process B? Explain.
You need to raise the temperature of a gas by 10°C. To use the least amount of heat energy, should you heat the gas at constant pressure or at constant volume? Explain.
What is the maximum mass of ethyl alcohol you could boil with 1000 J of heat, starting from 20 C?
You come into the lab one day and find a well-insulated thermos bottle containing of boiling liquid nitrogen. The remainder of the thermos has nitrogen gas at a pressure of atm. The gas and liquid are in thermal equilibrium. While waiting for lab to start, you notice a piece of iron on the table with " " written on it. Just for fun, you drop the iron into the thermos and seal the cap tightly so that no gas can escape. After a few seconds have passed, what is the pressure inside the thermos? The density of liquid nitrogen is .
a. 100 J of heat energy are transferred to 20 g of mercury. By how much does the temperature increase?
b. How much heat is needed to raise the temperature of 20 g of water by the same amount?
What do you think about this solution?
We value your feedback to improve our textbook solutions.