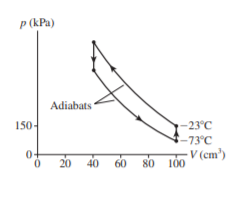
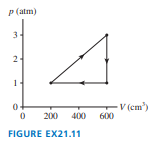
Chapter 21: Q. 25 (page 595)
A heat engine doesof work and exhaustsof waste heat during each cycle.
a. What is the engine's thermal efficiency?
b. If the cold-reservoir temperature is , what is the minimum possible temperature in of the hot reservoir?
Short Answer
a.Engine thermal efficiency is.
b.The minimum temperature of the heat reservoir is.