Chapter 21: Q. 68 (page 598)
In Problems 67 through 70 you are given the equation(s) used to solve a problem. For each of these, you are to
a. Write a realistic problem for which this is the correct equation(s).
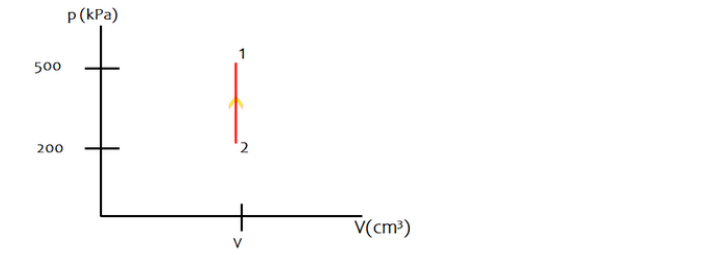
b. Draw a pVdiagram.
c. Finish the solution of the problem.
Short Answer
a) A realistic problem for which this is the correct equation is
"A gas in a hard container is at a temperature of 400oC and a pressure of 500 kPa. The container's internal pressure is decreased to 200kPa. Calculate the final temperature in degrees Celsius."
b) The required pV diagram is
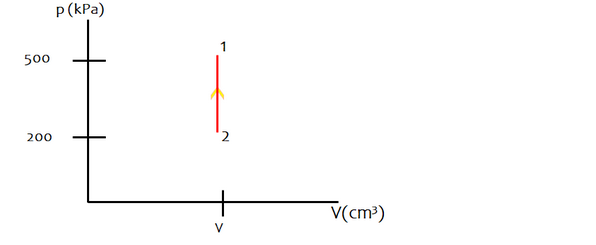
c) The solution of the problem is -4 oC