Chapter 21: Q. 24 (page 595)
At what cold-reservoir temperature inwould a Carnot engine with a hot-reservoir temperature of have an efficiency of
Short Answer
Cold-reservoir temperature for carnot engine is.
Learning Materials
Features
Discover
Chapter 21: Q. 24 (page 595)
At what cold-reservoir temperature inwould a Carnot engine with a hot-reservoir temperature of have an efficiency of
Cold-reservoir temperature for carnot engine is.
All the tools & learning materials you need for study success - in one app.
Get started for free
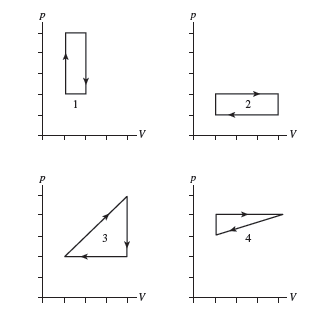
Rank in order, from largest to smallest, the amount of work done by the gas in each of the cycles shown in FIGURE Q21.2. Explain.
A heat engine uses a diatomic gas in a Brayton cycle. What is the engine's thermal efficiency if the gas volume is halved during the adiabatic compression?
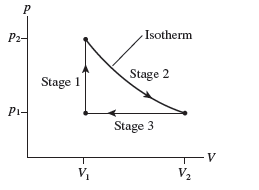
FIGURE Q21.4 shows the pV diagram of a heat engine. During which stage or stages is (a) heat added to the gas,(b) heat removed from the gas, (c) work done on the gas, and (d) work done by the gas?
A Carnot engine operating between energy reservoirs at temperatures and produces a power output of . What are (a) the thermal efficiency of this engine, (b) the rate of heat input, in and (c) the rate of heat output, in ?
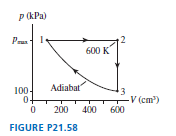
A heat engine using a monatomic gas follows the cycle shown in FIGURE P.
a. Find , and for each process in the cycle. Display your results in a table.
b. What is the thermal efficiency of this heat engine?
What do you think about this solution?
We value your feedback to improve our textbook solutions.