Chapter 21: Q. 2 (page 593)
Rank in order, from largest to smallest, the amount of work done by the gas in each of the cycles shown in FIGURE Q21.2. Explain.
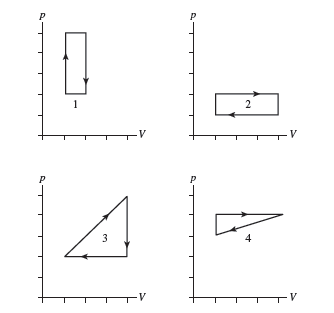
Short Answer
Rank in order is
Learning Materials
Features
Discover
Chapter 21: Q. 2 (page 593)
Rank in order, from largest to smallest, the amount of work done by the gas in each of the cycles shown in FIGURE Q21.2. Explain.

Rank in order is
All the tools & learning materials you need for study success - in one app.
Get started for free
A Carnot heat engine operates between reservoirs at and . If the engine extracts of energy from the hot reservoir per cycle, how many cycles will it take to lift a mass a height of ?
Engineers testing the efficiency of an electric generator gradually vary the temperature of the hot steam used to power it while leaving the temperature of the cooling water at a constant . They find that the generator’s efficiency increases at a rate at steam temperatures near . What is the ratio of the generator’s efficiency to the efficiency of a Carnot engine?
The coefficient of performance of a refrigerator is . The refrigerator's compressor uses of electric power and is efficient at converting electric power into work. What are (a) the rate at which heat energy is removed from inside the refrigerator and (b) the rate at which heat energy is exhausted into the room?
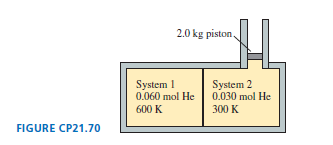
FIGURE CPshows two insulated compartments separated by a thin wall. The left side contains of helium at an initial temperature of and the right side contains of helium at an initial temperature of . The compartment on the right is attached to a vertical cylinder, above which the air pressure is . A -diameter, piston can slide without friction up and down the cylinder. Neither the cylinder diameter nor the volumes of the compartments are known.
a. What is the final temperature?
b. How much heat is transferred from the left side to the right side?
c. How high is the piston lifted due to this heat transfer?
d. What fraction of the heat is converted into work?
The power output of a car engine running at is . How much (a) work is done and (b) heat is exhausted per cycle if the engine's thermal efficiency is ? Give your answers in.
What do you think about this solution?
We value your feedback to improve our textbook solutions.