Chapter 31: Q9E (page 1045)
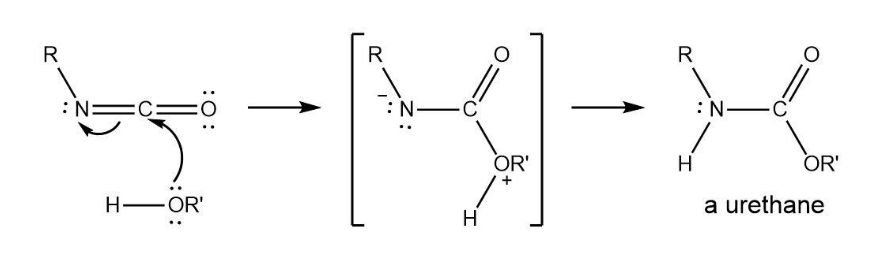
Show the mechanism of the nucleophilic addition reaction of an alcohol with an isocyanate to yield a urethane.
Short Answer
Structure of urethane followed as:
Learning Materials
Features
Discover
Chapter 31: Q9E (page 1045)
Show the mechanism of the nucleophilic addition reaction of an alcohol with an isocyanate to yield a urethane.
Structure of urethane followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
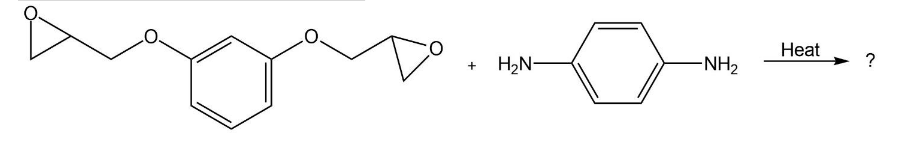
Show the structure of the polymer that results from heating the following diepoxide and diamine:
Vinylidene chloride, H2C = CHCl, does not polymerize in isotactic, syndiotactic, and atactic forms. Explain.
Melmac, a thermosetting resin often used to make plastic dishes, is prepared by heating melamine with formaldehyde. Look at the structure of Bakelite shown in Section 31-6, and then propose a structure for Melmac.
Polystyrene is produced commercially by the reaction of styrene with butyllithium as an anionic initiator. Using resonance structures, explain how the chain-carrying intermediate is stabilized.
Order the following monomers concerning their expected reactivity toward anionic polymerization, and explain your answer:
H2C = CHCH3, H2C = CHC N, H2C = CH-C6H5
What do you think about this solution?
We value your feedback to improve our textbook solutions.