Chapter 31: Q3E (page 1039)
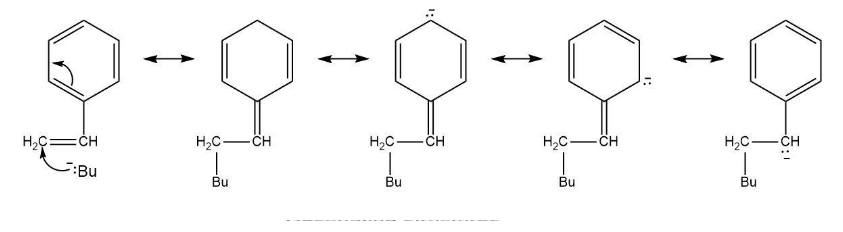
Polystyrene is produced commercially by the reaction of styrene with butyllithium as an anionic initiator. Using resonance structures, explain how the chain-carrying intermediate is stabilized.
Short Answer
Resonating structures is:
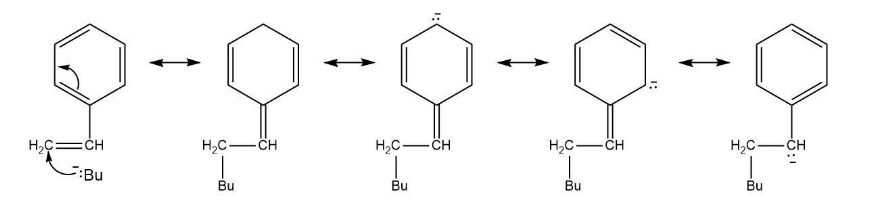
Intermediate are stabilized by resonance involving phenyl group.