Chapter 31: Q2E (page 1039)
Order the following monomers concerning their expected reactivity toward anionic polymerization, and explain your answer:
H2C = CHCH3, H2C = CHC N, H2C = CH-C6H5
Short Answer
Reactivity order follows as:
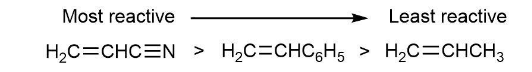
Learning Materials
Features
Discover
Chapter 31: Q2E (page 1039)
Order the following monomers concerning their expected reactivity toward anionic polymerization, and explain your answer:
H2C = CHCH3, H2C = CHC N, H2C = CH-C6H5
Reactivity order follows as:
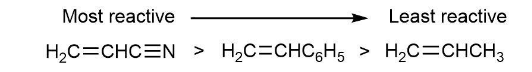
All the tools & learning materials you need for study success - in one app.
Get started for free
Nomex, a polyamide used in such applications as fire-retardant clothing, is prepared by reaction of 1,3-benzenediamine with 1,3-benzenedicarbonyl chloride. Show the structure of Nomex.
Melmac, a thermosetting resin often used to make plastic dishes, is prepared by heating melamine with formaldehyde. Look at the structure of Bakelite shown in Section 31-6, and then propose a structure for Melmac.
1,3-Cyclopentadiene undergoes thermal polymerization to yield a polymer that has no double bonds in the chain. Upon strong heating, the polymer breaks down to generate cyclopentadiene. Propose a structure of the polymer.
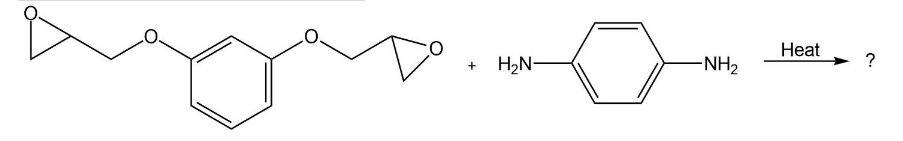
Show the structure of the polymer that results from heating the following diepoxide and diamine:
Look at the structures of Vestenamer and Norsorex and show how they might be made by olefin metathesis polymerization.
What do you think about this solution?
We value your feedback to improve our textbook solutions.