Chapter 13: Q13-34E (page 419)
How many types of non-equivalent protons are present in each of the following molecules?
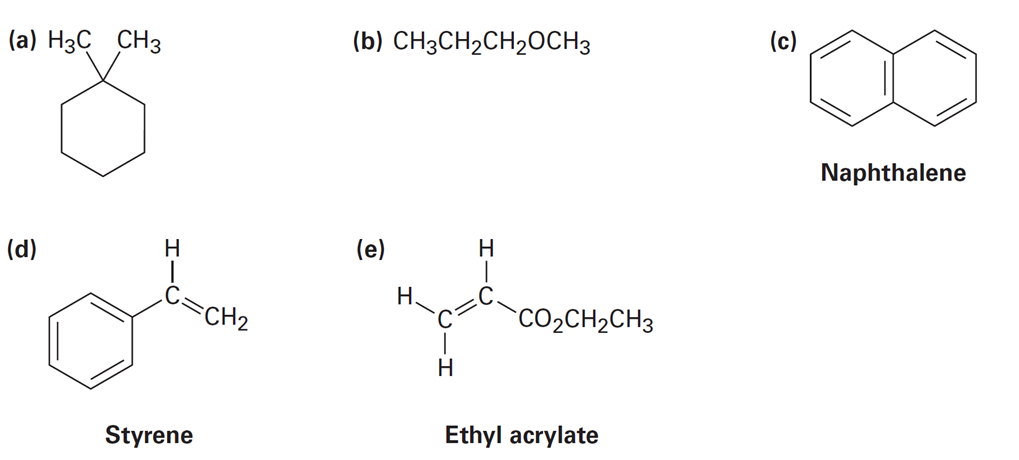
Short Answer
- 4
- 4
- 2
- 6
- 5
Learning Materials
Features
Discover
Chapter 13: Q13-34E (page 419)
How many types of non-equivalent protons are present in each of the following molecules?

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Predict the number of carbon resonance lines you would expect in the C- 13 NMR spectra of the following compounds:
(a) Methylcyclopentane
(b) 1- Methylcyclohexane
(c) 1,2- Dimethylbenzene
(d) 2- Methyl-2-butene
(e)
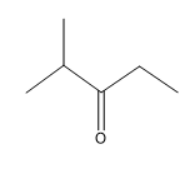
(f)
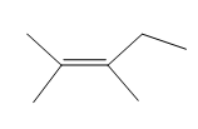
Assume that you have a compound with the formula C3H6O .
(a) How many double bonds and/or rings does your compound
contain?
(b) Propose as many structures as you can that fit the molecular
formula.
(c) If your compound shows an infrared absorption peak at 1715cm-1,
what functional group does it have?
(d) If your compound shows a single 1H NMR absorption peak at 2.1,
what is its structure?
Compound D is isomeric with compound C (Problem 13-61) and has the following NMR spectral data. Propose a structure.
Compound D Broadband-decoupled NMR: 9.7, 29.9, 74.4, 114.4, 141.4 d DEPT-90: 74.4, 141.4 d DEPT-135: positive peaks at 9.7, 74.4, 141.4 d; negative peaks at 29.9, 114.4 d
Propose structures for the two compounds whose1H NMR spectra are
shown.
(a) C4H9Br
(b) C4H8CI2
How many 13C NMR absorptions would you expect for cis-1,3-dimethylcyclohexane?For trans-1,3-dimethylcyclohexane? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.