Chapter 12: Q48E (page 354)
Rank the double bonds below in terms of increasing stability:
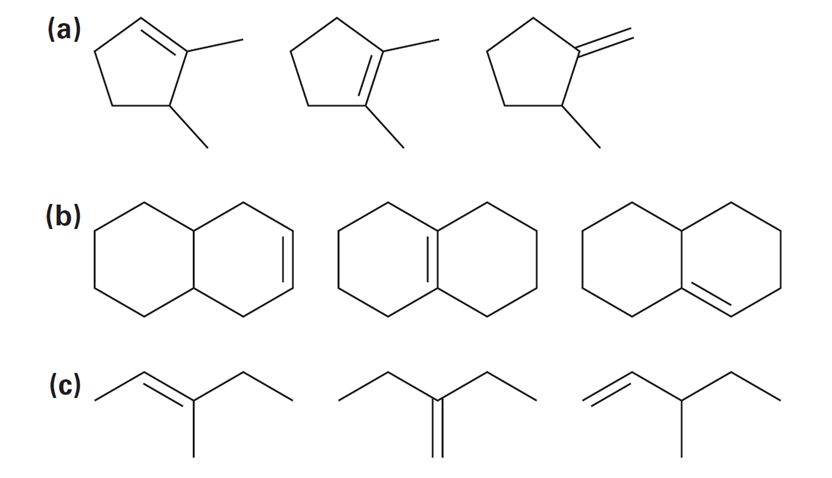
Short Answer
Learning Materials
Features
Discover
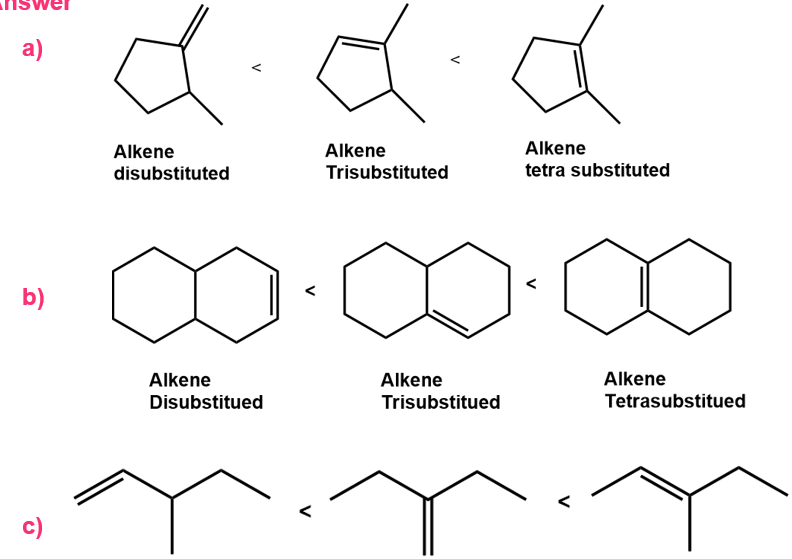
Chapter 12: Q48E (page 354)
Rank the double bonds below in terms of increasing stability:


All the tools & learning materials you need for study success - in one app.
Get started for free
Assume that you are carrying out the base-induced dehydrobromination of 3-Bromo-3-methyl pentane (Section 11-7) to yield an alkene. How could you use IR spectroscopy to tell which of three possible elimination products is formed, if one includes E/Z isomers?
Question: Why do you suppose accidental overlap of signals is much more common in1H NMR than in13C NMR?
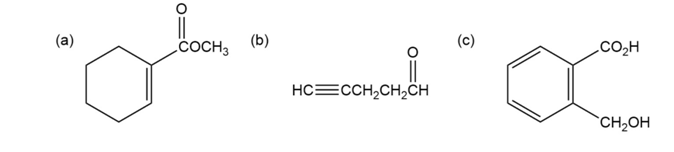
Question: At what approximate positions might the following compounds showIR absorptions?
(e)
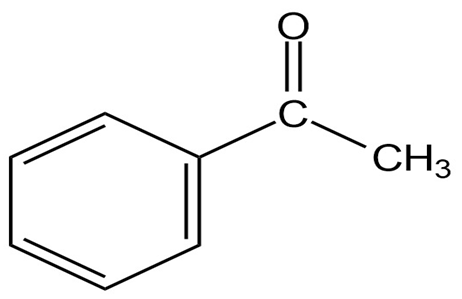
Which is stronger, the C=O bond in an ester () or the C=O bond in a saturated ketone ()? Explain
Where might the following compounds have IR absorptions?
What do you think about this solution?
We value your feedback to improve our textbook solutions.