Chapter 12: Q21E (page 354)
Predict the product(s) and show the complete electron-pushing mechanism for each of the following dissolving metal reductions.
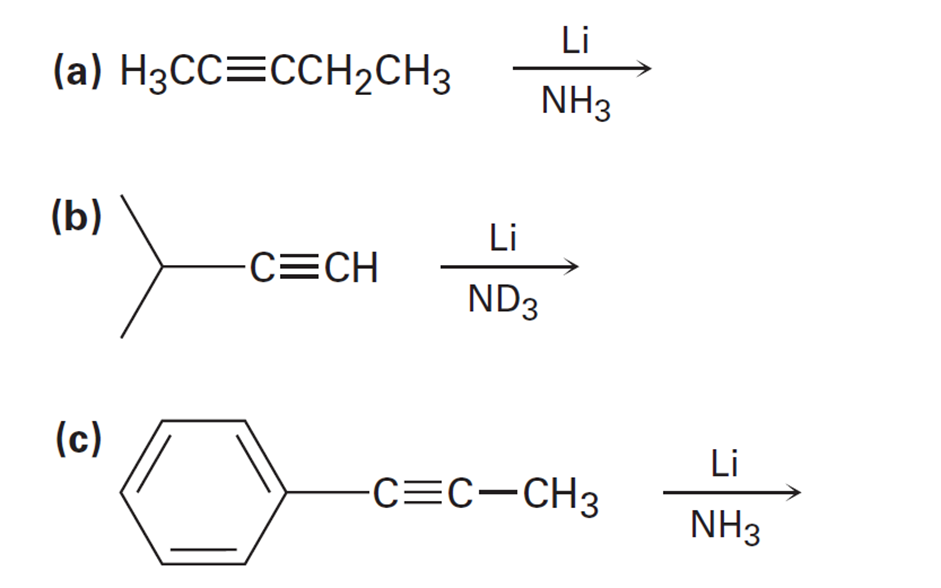
Short Answer
a).
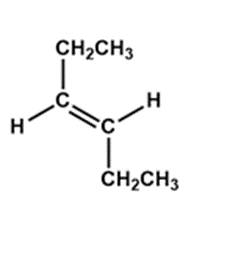
b).
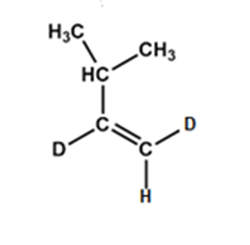
c).
Learning Materials
Features
Discover
Chapter 12: Q21E (page 354)
Predict the product(s) and show the complete electron-pushing mechanism for each of the following dissolving metal reductions.

a).

b).

c).

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Why do you suppose accidental overlap of signals is much more common in1H NMR than in13C NMR?
Carvone is an unsaturated ketone responsible for the odour of spearmint. If carvone hasin its mass spectrum and contains three double bonds and one ring, what is its molecular formula?
Question: 4-Methyl-2-pentanone and 3-methylpentanal are isomers. Explain how you could tell them apart, both by mass spectrometry and by infrared spectroscopy.
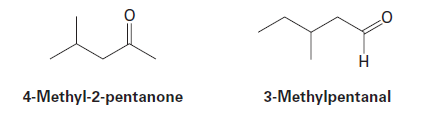
Question: Two infrared spectra are shown. One is the spectrum of cyclohexane,
and the other is the spectrum of cyclohexene. Identify them, and
explain your answer.
(a)
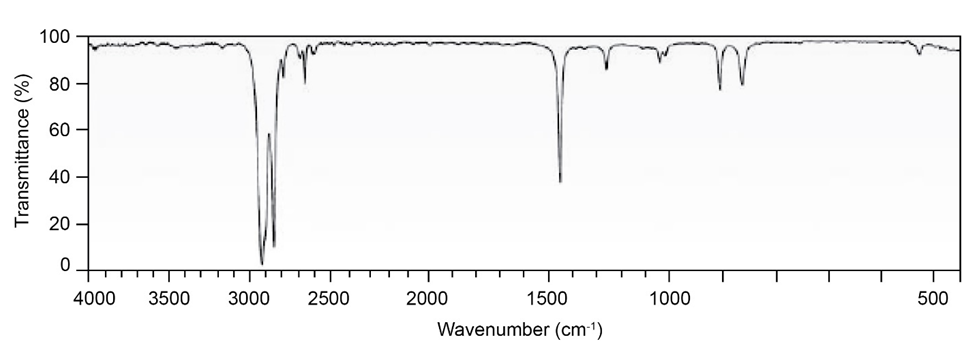
(b)
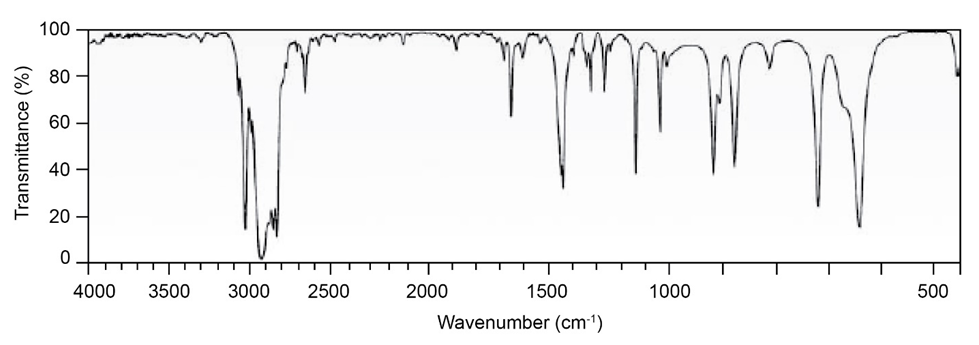
At what approximate positions might the following compounds showIR absorptions?
(d)
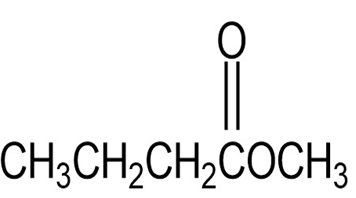
What do you think about this solution?
We value your feedback to improve our textbook solutions.