Chapter 12: Q-21-21-4P (page 354)
Rank the compounds in each of the following sets in order of their expected reactivity toward nucleophilic acyl substitution:
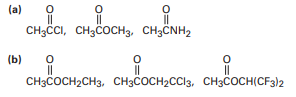
Short Answer
The order of reactivity towards nucleophilic acyl substitution:
(a)
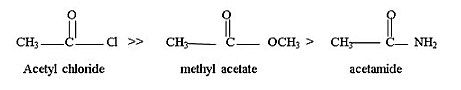
(b)
Learning Materials
Features
Discover
Chapter 12: Q-21-21-4P (page 354)
Rank the compounds in each of the following sets in order of their expected reactivity toward nucleophilic acyl substitution:

The order of reactivity towards nucleophilic acyl substitution:
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What functional groups might the following molecules contain?
(a) A compound with a strong absorption at 1710
(b) A compound with a strong absorption at 1540
(c) A compound with strong absorptions at 1720 and 2500 to 3100
Question: Grignard reagents undergo a general and very useful reaction with
ketones. Methylmagnesium bromide, for example, reacts with cyclohexanoneto yield a product with the formula . What is the structure of this product if it has an IR absorption at ?
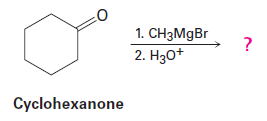
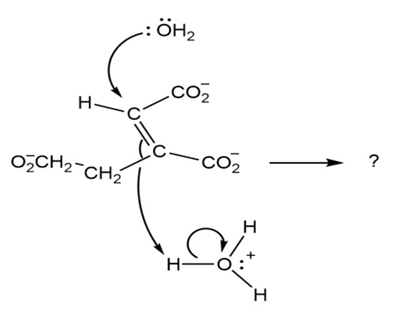
Predict the products of the following polar reaction, a step in the citric acid cycle for food metabolism, by interpreting the flow of electrons indicated by the curved arrows:
Question: Halogenated compounds are particularly easy to identify by their mass
spectra because both chlorine and bromine occur naturally as mixtures
of two abundant isotopes. Recall that chlorine occurs as 35Cl (75.8%)
and 37C (24.2%); and bromine occurs as 79Br (50.7%) and 81Br (49.3%).
At what masses do the molecular ions occur for the following formulas?
What are the relative percentages of each molecular ion?
(a)Bromomethane, CH3Br
(b)1-Chlorohexane, C6H13Cl
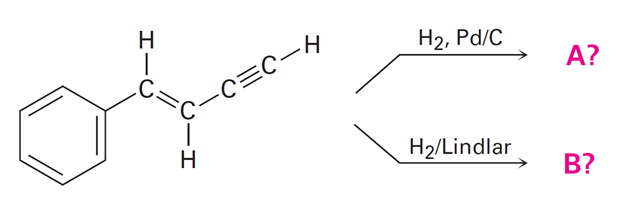
Predict the products of the following reactions:
What do you think about this solution?
We value your feedback to improve our textbook solutions.