Chapter 12: Q12-5P (page 371)
Which has higher energy, infrared radiation with or anX-ray with ? Radiation with or with ?
Short Answer
X-ray has more energy than infrared radiation.
Radiation with has more energy than
Learning Materials
Features
Discover
Chapter 12: Q12-5P (page 371)
Which has higher energy, infrared radiation with or anX-ray with ? Radiation with or with ?
X-ray has more energy than infrared radiation.
Radiation with has more energy than
All the tools & learning materials you need for study success - in one app.
Get started for free
Which is stronger, the C=O bond in an ester () or the C=O bond in a saturated ketone ()? Explain
Identify the functional groups in each of the following molecules:
(a) Methionine, an amino acid:
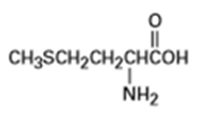
(b)Ibuprofen, a pain reliever:
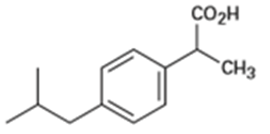
(c) Capsaicin, the pungent substance in chili peppers:
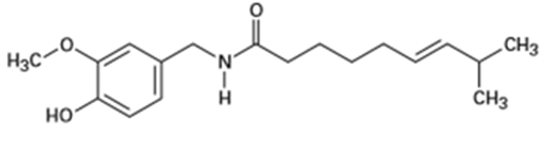
Question: At what approximate positions might the following compounds showIR absorptions?
(f)
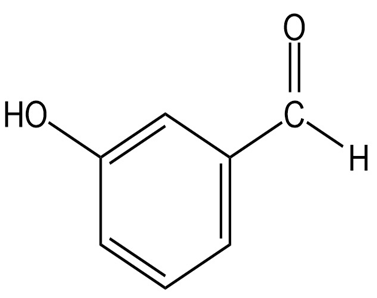
Question: At what approximate positions might the following compounds showIR absorptions?
(e)
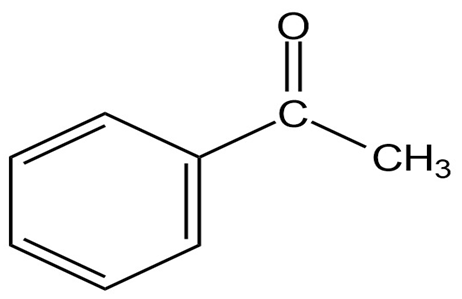
How would you prepare the following substances, starting from any
compounds having four carbons or fewer?
What do you think about this solution?
We value your feedback to improve our textbook solutions.