Chapter 12: Q12-46 (page 354)
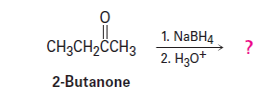
Ketones undergo a reduction when treated with sodium borohydride, . What is the structure of the compound produced by reaction of 2-butanone with if it has an IR absorption at and in the mass spectrum?
Short Answer
Learning Materials
Features
Discover
Chapter 12: Q12-46 (page 354)
Ketones undergo a reduction when treated with sodium borohydride, . What is the structure of the compound produced by reaction of 2-butanone with if it has an IR absorption at and in the mass spectrum?


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: At what approximate positions might the following compounds show
IR absorptions?
(c)
How might you use IR spectroscopy to distinguish between the followingpairs of isomers?
(a) CH3 CH2 OH and CH3 OCH3
(b) Cyclohexane and 1 - hexene
(c) CH3 CH2 CO2 H and HOCH2 CH2 CHO
Predict the products of the following reactions:
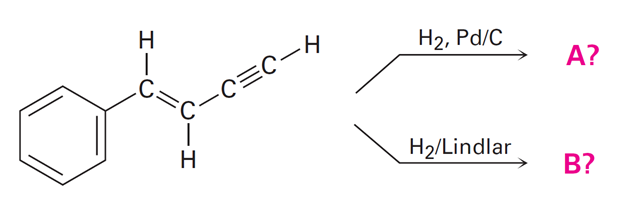
Question: Grignard reagents undergo a general and very useful reaction with
ketones. Methylmagnesium bromide, for example, reacts with cyclohexanoneto yield a product with the formula . What is the structure of this product if it has an IR absorption at ?
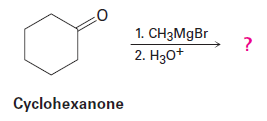
Amino acids can be prepared by reaction of alkyl halides with diethyl acetamidomalonate, followed by heating the initial alkylation product with aqueous HCl. Show how you would prepare alanine, , one of the twenty amino acids found in proteins, and propose a mechanism for acid-catalyzed conversion of the initial alkylation product to the amino acid.

What do you think about this solution?
We value your feedback to improve our textbook solutions.