Chapter 12: Q12-47E (page 354)
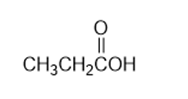
Nitriles, , undergo a hydrolysis reaction when heated withaqueous acid. What is the structure of the compound produced by hydrolysis of propanenitrile, , if it has IR absorptions from and at , and has ?
Short Answer
Learning Materials
Features
Discover
Chapter 12: Q12-47E (page 354)
Nitriles, , undergo a hydrolysis reaction when heated withaqueous acid. What is the structure of the compound produced by hydrolysis of propanenitrile, , if it has IR absorptions from and at , and has ?

All the tools & learning materials you need for study success - in one app.
Get started for free
Assume that you are carrying out the base-induced dehydrobromination of 3-Bromo-3-methyl pentane (Section 11-7) to yield an alkene. How could you use IR spectroscopy to tell which of three possible elimination products is formed, if one includes E/Z isomers?
Question: The nitrogen ruleof mass spectrometry says that a compound containing an odd number of nitrogen’s has an odd-numbered molecular ion.
Conversely, a compound containing an even number of nitrogen’s has
an even-numbered M+peak. Explain.
What functional groups might the following molecules contain?
(a) A compound with a strong absorption at 1710 cm-1
(b) A compound with a strong absorption at 1540 cm-1
(c) A compound with strong absorptions at 1720 cm-1 and 2500 to 3100cm-1
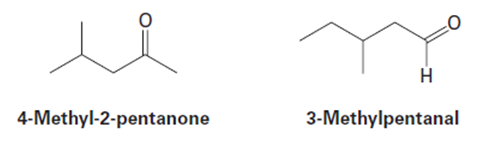
4-Methyl-2-pentanone and 3-methylpentanal are isomers. Explain how
you could tell them apart, both by mass spectrometry and by infrared
spectroscopy.
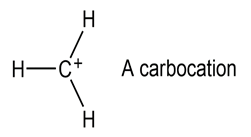
Most stable organic species have tetravalent carbon atoms, but species with trivalent carbon atoms also exist. Carbocations are one such class of compounds.
(a) How many valence electrons does the positively charged carbon atom have?
(b) What hybridization do you expect this carbon atom to have?
(c) What geometry is the carbocation likely to have?
What do you think about this solution?
We value your feedback to improve our textbook solutions.