Chapter 12: Q 42 E (page 385)
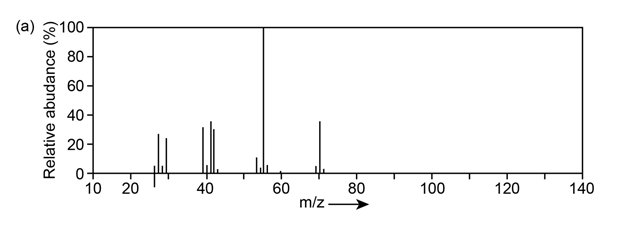
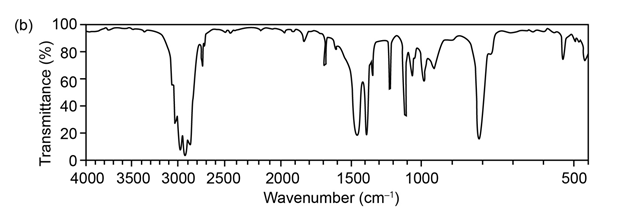
Question: The mass spectrum (a) and the infrared spectrum (b) of another unknown hydrocarbon are shown. Propose as many structures as you can.
Short Answer
Learning Materials
Features
Discover
Chapter 12: Q 42 E (page 385)
Question: The mass spectrum (a) and the infrared spectrum (b) of another unknown hydrocarbon are shown. Propose as many structures as you can.



All the tools & learning materials you need for study success - in one app.
Get started for free
At what approximate positions might the following compounds showIR absorptions?
(d)
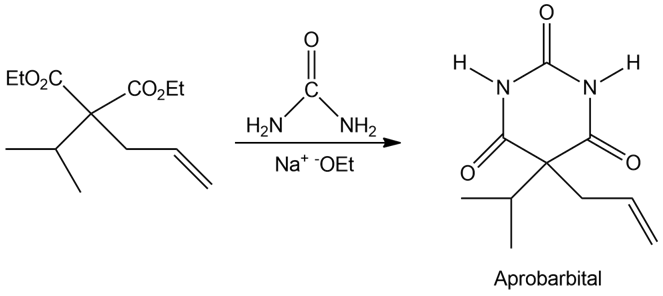
Aprobarbital, a barbiturate once used in treating insomnia, is synthesized in three steps from diethyl malonate. Show how you would synthesize the necessary dialkylated intermediate, and then propose a mechanism for the reaction of this intermediate with urea to give aprobarbital.
Question: Why do you suppose accidental overlap of signals is much more common in1H NMR than in13C NMR?
Predict the product and provide the complete electron-pushing mechanism for the following two-step synthetic processes:

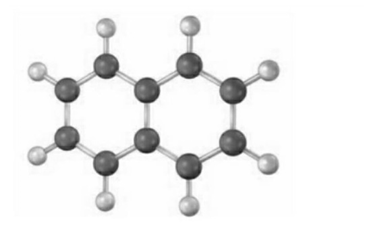
Fill in the multiple bonds in the following model of naphthalene, (gray 5 C, ivory 5 H). How many resonance structures does naphthalene have? Draw them.
What do you think about this solution?
We value your feedback to improve our textbook solutions.