Chapter 12: Structure Determination: Mass Spectroscopy and Infrared Spectroscopy
Q35f E
Question: At what approximate positions might the following compounds showIR absorptions?
(f)
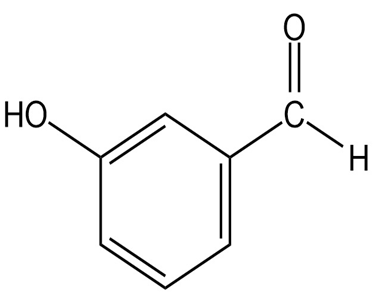
Q37E
Identify all the acidic hydrogens () in the following molecules:
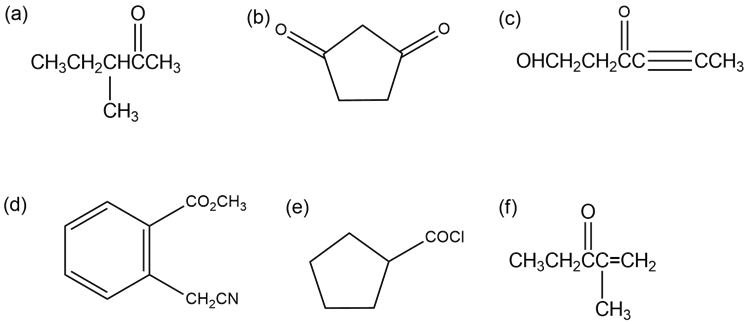
Q40E
Base treatment of the following α,β-unsaturated carbonyl compound yields an anion by removal of H+ from thecarbon. Why are hydrogens on thecarbon atom acidic?
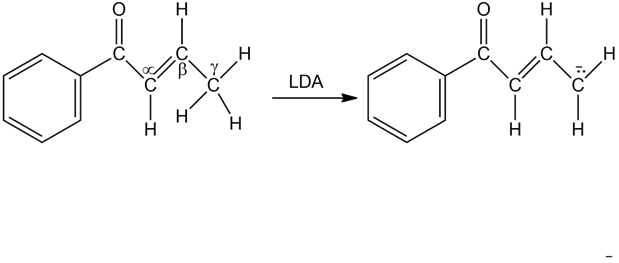
Q41E
Although the Diels–Alder reaction generally occurs between an electron rich diene and an electron-deficient dienophile, it is also possible to
have inverse-demand Diels–Alder reactions between suitable electrondeficient
conjugated double bonds and electron-rich alkenes. These reactions are particularly useful because they allow for the incorporation of heteroatoms into the new six-membered ring. Predict the products of each inverse-demand Diels–Alder reaction below. Be sure your products reflect the correct stereochemistry. If more than one region isomer is possible, draw both.
(a)
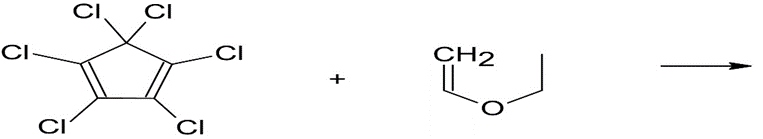
(b)
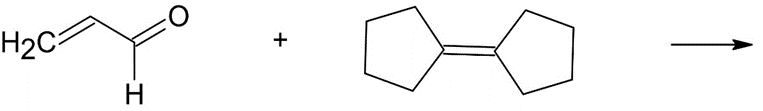
(c)

Q41E
Treatment of 1-phenyl-2-propenone with a strong base such as LDA does not yield an anion, even though it contains a hydrogen on the carbon atom next to the carbonyl group. Explain.
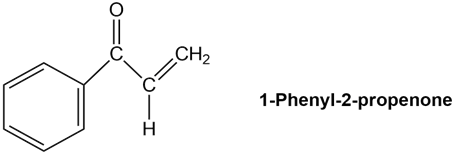
Q 41 E
Question:The mass spectrum (a) and the infrared spectrum (b) of an unknown hydrocarbon are shown. Propose as many structures as you can.
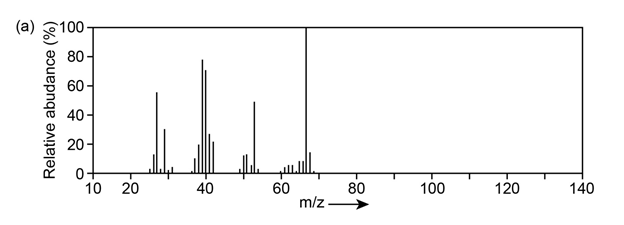
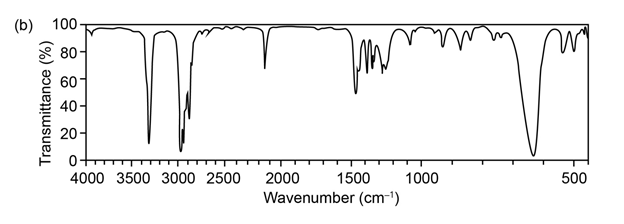
Q 42 E
Question: The mass spectrum (a) and the infrared spectrum (b) of another unknown hydrocarbon are shown. Propose as many structures as you can.

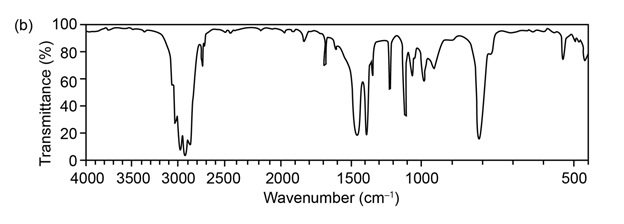
Q42 E
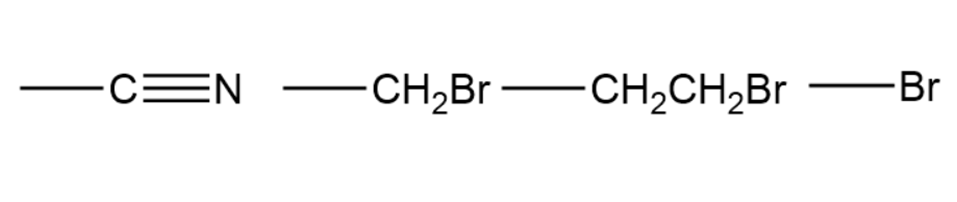
Assign Cahn–Ingold–Prelog rankings to the following sets of substituents
a.
b.
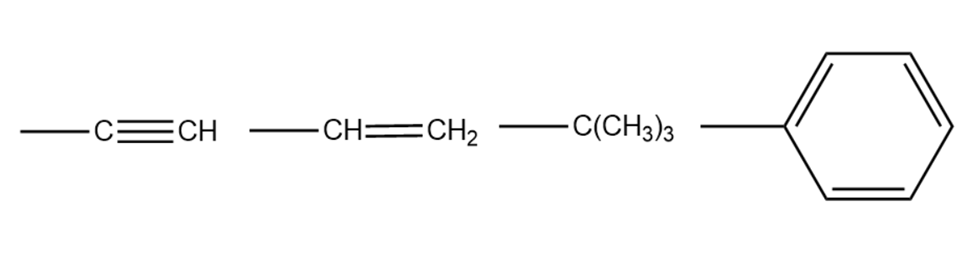
c.
d.,
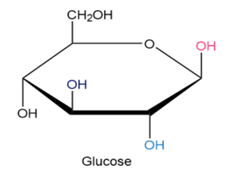
Q4-32E
Identify each pair of relationships among the groups in glucose (pink–blue, pink–dark blue, pink–black, blue–dark blue, blue-black, dark blue-black) as cis or trans.
Q43E
Propose structures for compounds that meet the following descriptions:
- An optically active compound C5H10Owith an IR absorption at 1730 cm-1.
- A non-optically active compound C5H9N with an IR absorption at 2215cm-1.
