Chapter 12: Q37E (page 354)
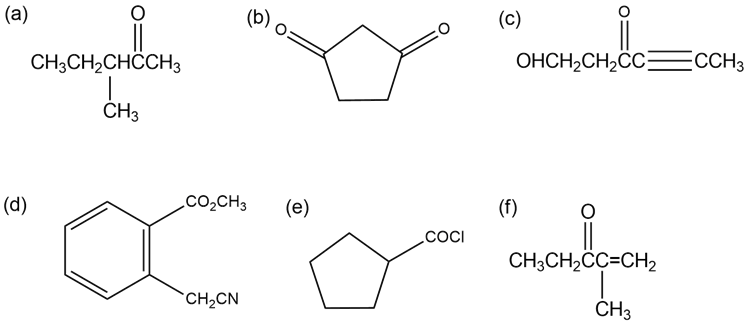
Identify all the acidic hydrogens () in the following molecules:
Short Answer
 Acidic hydrogens in (a)
Acidic hydrogens in (a)
 Acidic hydrogens in (b)
Acidic hydrogens in (b)
 Acidic hydrogens in (c)
Acidic hydrogens in (c)
 Acidic hydrogens in (d)
Acidic hydrogens in (d)
 Acidic hydrogen in (e)
Acidic hydrogen in (e)
 Acidic hydrogen in (f)
Acidic hydrogen in (f)
Learning Materials
Features
Discover
Chapter 12: Q37E (page 354)
Identify all the acidic hydrogens () in the following molecules:

 Acidic hydrogens in (a)
Acidic hydrogens in (a)
 Acidic hydrogens in (b)
Acidic hydrogens in (b)
 Acidic hydrogens in (c)
Acidic hydrogens in (c)
 Acidic hydrogens in (d)
Acidic hydrogens in (d)
 Acidic hydrogen in (e)
Acidic hydrogen in (e)
 Acidic hydrogen in (f)
Acidic hydrogen in (f)
All the tools & learning materials you need for study success - in one app.
Get started for free
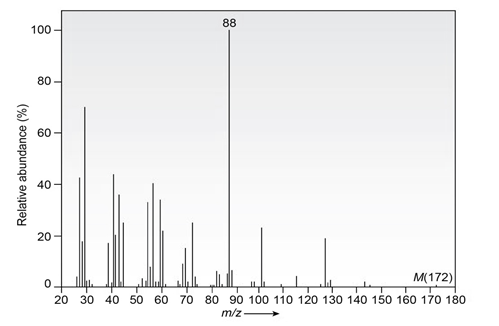
Question: The infrared spectrum of the compound with the mass spectrum shown below lacks any significant absorption above . There is a prominent peak near and another strong peak near .Propose a structure consistent with the data.
Question: What functional groups might the following molecules contain?
(a) A compound with a strong absorption at 1710
(b) A compound with a strong absorption at 1540
(c) A compound with strong absorptions at 1720 and 2500 to 3100
Name the following alkynes, and predict the products of their reaction
With:
(1) in the presence of a Lindlar’s catalyst
(2) in the presence of
Question: How might you use IR spectroscopy to distinguish between the following pairs of isomers?
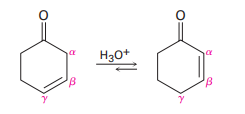
Nonconjugated ,-unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated ,-unsaturated isomers. Propose a mechanism for this isomerization.
What do you think about this solution?
We value your feedback to improve our textbook solutions.