Chapter 11: Q60E (page 350)
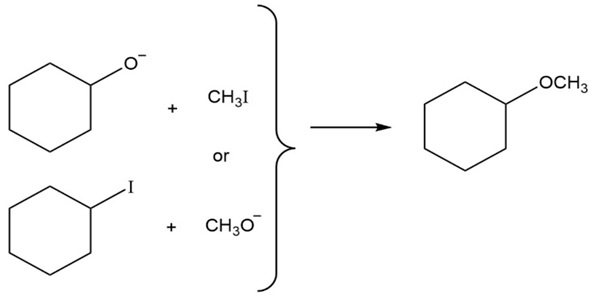
Ethers can often be prepared by SN2 reaction of alkoxide ions, ,with alkyl halides. Suppose you wanted to prepare cyclohexyl methylether. Which of the two possible routes shown below would youchoose? Explain.
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q60E (page 350)
Ethers can often be prepared by SN2 reaction of alkoxide ions, ,with alkyl halides. Suppose you wanted to prepare cyclohexyl methylether. Which of the two possible routes shown below would youchoose? Explain.


All the tools & learning materials you need for study success - in one app.
Get started for free
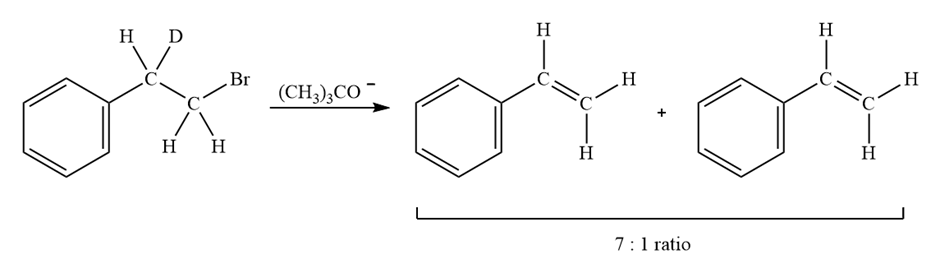
Treatment of 1-bromo-2-deuterio-2-phenylethane with strong base leads to a mixture of deuterated and nondeuterated phenylethylenes in an approximately 7;1 ratio. Explain
Which compound in each of the following pairs will react faster in an SN2reaction with OH-?
(b) CH3CH2Iin ethanol or dimethyl sulfoxide
Which reactant in each of the following pairs is more nucleophilic? Explain.
(e) I- or Cl-
Question: (R)-2-Bromooctane undergoes racemization to give ()-2-bromooctane when treated with NaBr in dimethyl sulfoxide. Explain.
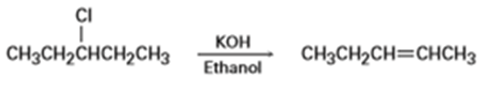
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB or E2:
b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.