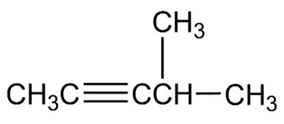
Chapter 11: Q55A (page 350)
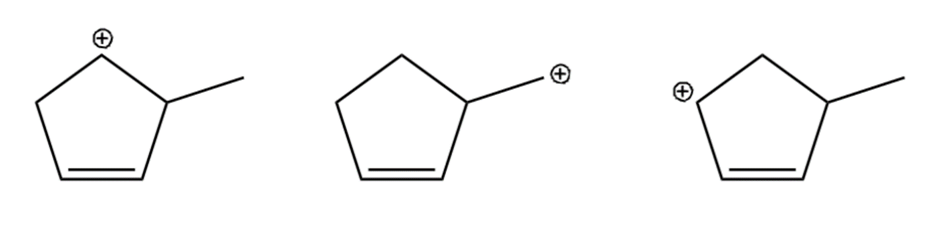
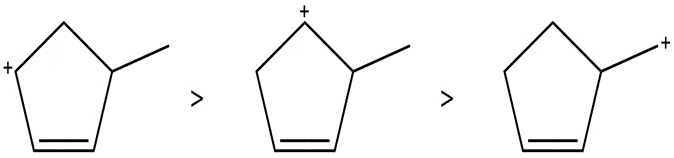
Question: Arrange the carbocations below in order of increasing stability.
a.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 11: Q55A (page 350)
Question: Arrange the carbocations below in order of increasing stability.
a.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Order each of the following sets of compounds with respect to reactivity:
b. (CH3)3 CCI (CH3)3 CBr (CH3)3 COH
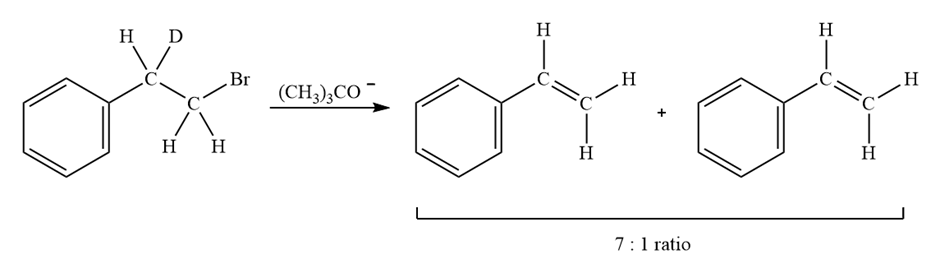
Treatment of 1-bromo-2-deuterio-2-phenylethane with strong base leads to a mixture of deuterated and nondeuterated phenylethylenes in an approximately 7;1 ratio. Explain
Which compound in each of the following pairs will react faster in anreaction with?
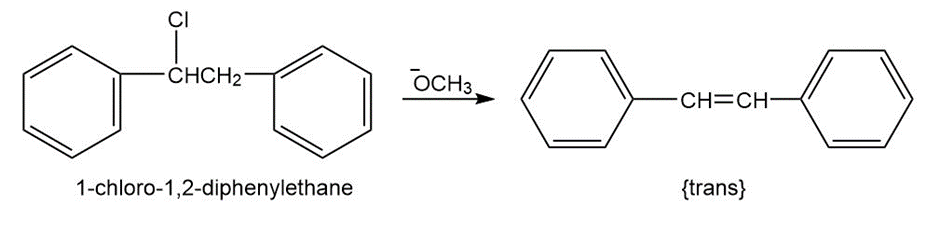
Question: 1-Chloro-1,2-diphenylethane can undergo E2 elimination to give either cis- or trans-1,2-diphenylethylene (stilbene). Draw Newman projections of the reactive conformations leading to both possible products, and suggest a reason why the trans alkene is the major product.
How might you prepare each of the following molecules using a nucleophilic substitution reaction at some step?
(a)
What do you think about this solution?
We value your feedback to improve our textbook solutions.