Chapter 11: 41aE (page 350)
Which compound in each of the following pairs will react faster in anreaction with?
Short Answer
a.
Learning Materials
Features
Discover
Chapter 11: 41aE (page 350)
Which compound in each of the following pairs will react faster in anreaction with?
a.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actual product.
b.
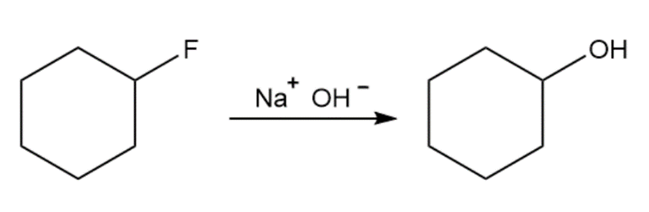
What effect would you expect the following changes to have on the rate of the reaction of ethanol with 2-iodo-2-methyl butane?
(b) The concentration of the ethanol is halved by adding diethyl ether as an inert solvent.
Which reaction in each of the following pairs would you expect to be faster?
(a) Thedisplacement by
Question: Arrange the carbocations below, in order of increasing stability:
b.
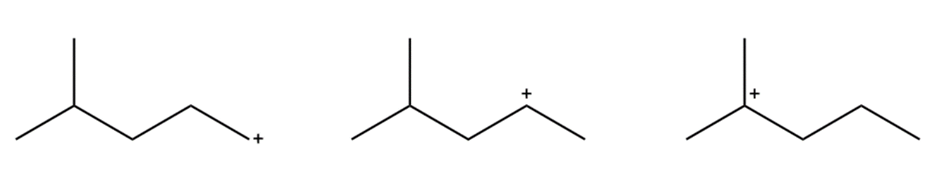
Question: Arrange the carbocations below in order of increasing stability.
a.
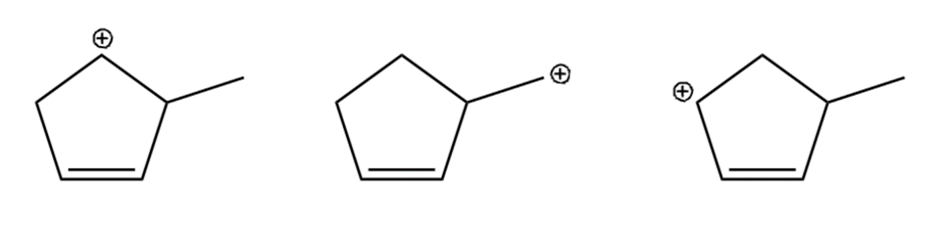
What do you think about this solution?
We value your feedback to improve our textbook solutions.