Chapter 8: Q8-56 E (page 262)
Poly (vinyl pyrrolidone), prepared from N-vinylpyrrolidone, is used in cosmetics and as a synthetic substitute for blood. Draw a representative segment of the polymer.
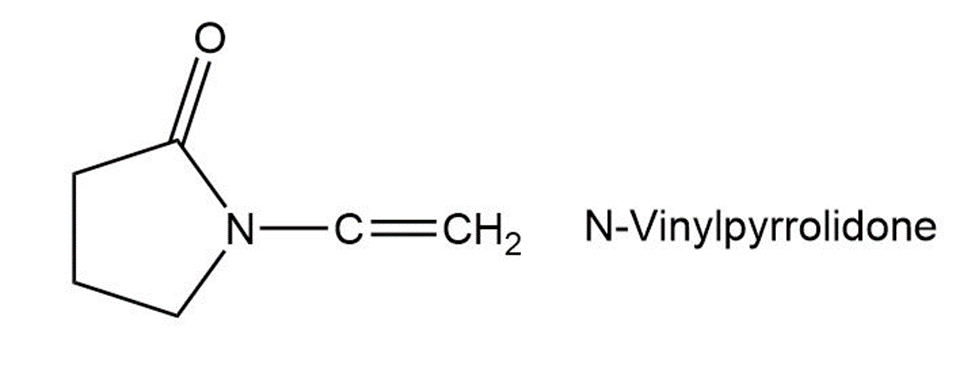
Short Answer
Answer
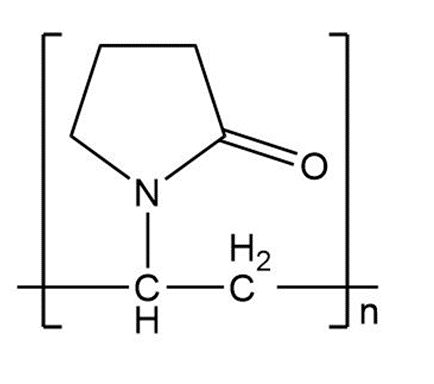
Learning Materials
Features
Discover
Chapter 8: Q8-56 E (page 262)
Poly (vinyl pyrrolidone), prepared from N-vinylpyrrolidone, is used in cosmetics and as a synthetic substitute for blood. Draw a representative segment of the polymer.
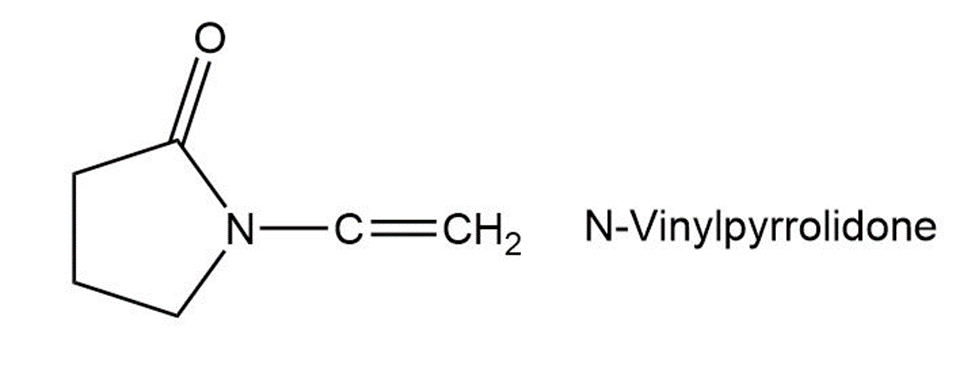
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
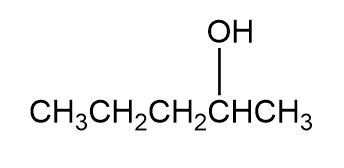
Question: Which of the following alcohols could notbe made selectively by hydroboration–oxidation of an alkene? Explain.
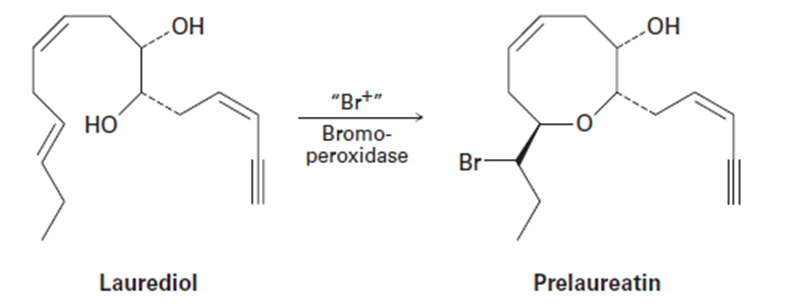
Isolated from marine algae, prelaureatin is thought to be biosynthesized from laurediol by the following route. Propose a mechanism
One problem with elimination reactions is that mixtures of products are often formed. For example, treatment of 2-bromo-2-methylbutane with KOH in ethanol yields a mixture of two alkene products. What are their likely structures?
Compound A, , was found to be an optically active alcohol. Despite its apparent unsaturation, no hydrogen was absorbed on catalytic reduction over a palladium catalyst. On treatment of A with dilute sulfuric acid, dehydration occurred and an optically inactive alkene B,, was the major product. Alkene B, on ozonolysis, gave two products. One product was identified as propanal,. Compound C, the other product, was shown to be a ketone,. How many degrees of unsaturation does A have? Write the reactions, and identify A, B, and C.
Question:How would you carry out the following transformations? Tell what
reagents you would use in each case.
(b)
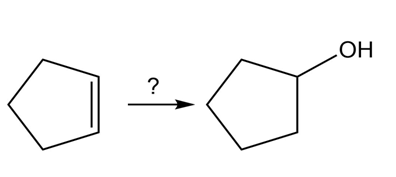
What do you think about this solution?
We value your feedback to improve our textbook solutions.