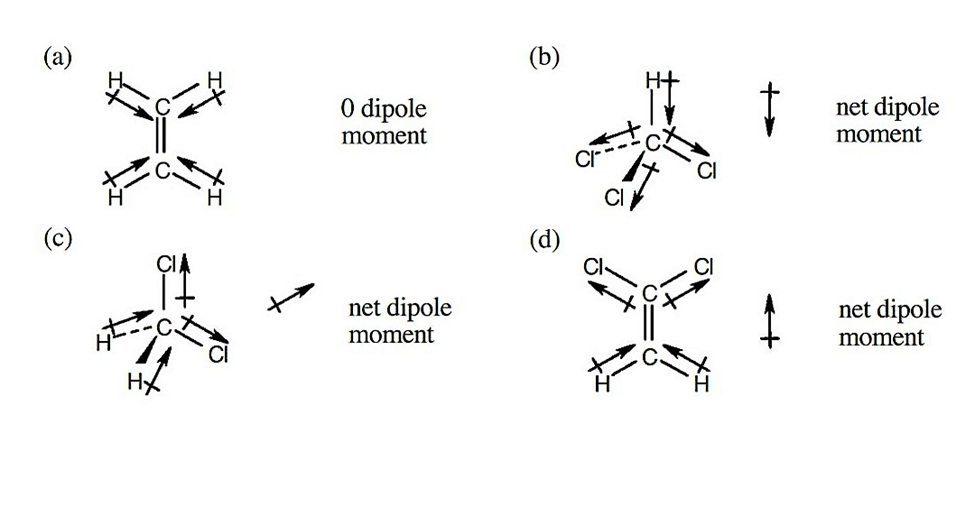
Chapter 2: Q6P. (page 33)
Make three-dimensional drawings of the following molecules, and predictwhether each has a dipole moment. If you expect a dipole moment, show itsdirection.
Short Answer
Learning Materials
Features
Discover
Chapter 2: Q6P. (page 33)
Make three-dimensional drawings of the following molecules, and predictwhether each has a dipole moment. If you expect a dipole moment, show itsdirection.

All the tools & learning materials you need for study success - in one app.
Get started for free
Calculate values from the following ’s:
(a)Acetone, =19.3 (b)Formic acid, =3.75
Ethylene glycol,, may look nonpolar when drawn, but an
internal hydrogen bond results in an electric dipole moment. Explain.
Question:Phosgene, Cl2C=O , has a smaller dipole moment than formaldehyde,H2C=O , even though it contains electronegative chlorine atoms in place of hydrogen. Explain.
Question: What is the Ka of HCN if its pKa=9.31?
Use the table in Appendix B to determine in which direction the
Equilibrium is favored.
(b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.