Chapter 4: Q-4-4-4E (page 94)
Name the following substances, including the cis- or trans- prefix:
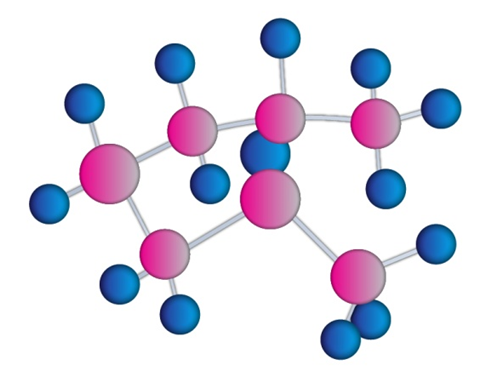
a)
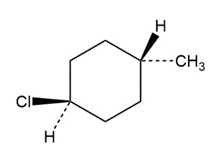
b)
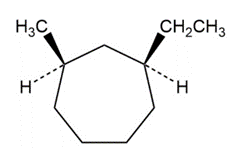
Short Answer
a) Trans-1chloro 4-methyl cyclohexane
b) Cis-1-ethyl,3-methyl cyclohexane
Learning Materials
Features
Discover
Chapter 4: Q-4-4-4E (page 94)
Name the following substances, including the cis- or trans- prefix:
a)

b)

a) Trans-1chloro 4-methyl cyclohexane
b) Cis-1-ethyl,3-methyl cyclohexane
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw three isomers of trans-1, 2-dichlorocyclobutane, and label them as either constitutional isomers or stereoisomers.
How many eclipsing interactions would be present if cyclopentanewere planar? Assuming an energy cost of 4.0 kJ/mol for each eclipsing interaction,how much torsional strain would planar cyclopentane have? Since themeasured total strain of cyclopentane is 26 kJ/mol, how much of the torsionalstrain is relieved by puckering?
Name the following cycloalkanes:
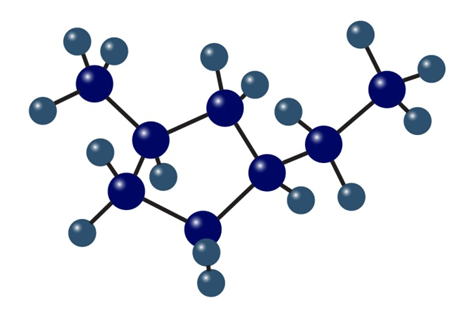
b. Name the following cycloalkanes:
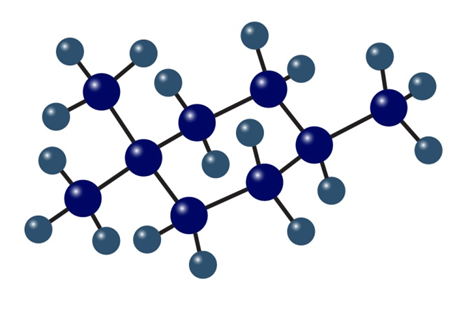
cis-1,2-Dimethylcyclopropane has more strain than trans-1,2-dimethylcyclopropane. How can you account for this difference? Which of the two compounds is more stable?
Name the following substances, including the cis- or trans- prefix (redbrown= Br):
What do you think about this solution?
We value your feedback to improve our textbook solutions.