Chapter 30: Q.30-12 (page 1033)
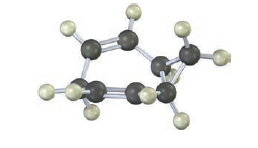
The 13C NMR spectrum of homotropilidene taken at room temperature shows only three peaks. Explain.
Short Answer
The13C NMR spectrum of homotropilidene taken at room temperature shows only three peaks can be explained.
Learning Materials
Features
Discover
Chapter 30: Q.30-12 (page 1033)
The 13C NMR spectrum of homotropilidene taken at room temperature shows only three peaks. Explain.

The13C NMR spectrum of homotropilidene taken at room temperature shows only three peaks can be explained.
All the tools & learning materials you need for study success - in one app.
Get started for free
(2E,4Z,6Z,8E)-2,4,6,8-Decatetraene has been cyclized to give 7,8-dimethyl-1,3,5-cyclooctatriene. Predict the manner of ring-closure- conrotatory or disrotatory for both thermal and photochemical reactions, and predict the stereochemistry of the product in each case.
Predict the product of the following pericyclic reaction. Is this [5,5] shift a suprafacial or an antarafacial process?
Which of the following reactions is more likely to occur? Explain.
Photolysis of the cis-cyclobutene isomer in Problem 30-35 yields cis-cyclododecaen-7-yne, but photolysis of the trans isomer yields trans-cyclododecaen-7-yne. Explain these results, and identify the type and stereochemistry of the pericyclic reaction.
Look at Figure 30-1, and tell which molecular orbital is the HOMO and whichis the LUMO for both ground and excited states of ethylene and 1,3-butadiene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.