Chapter 30: Q28E (page 1033)
Predict the product of the following pericyclic reaction. Is this [5,5] shift a suprafacial or an antarafacial process?
Short Answer
The product of the following pericyclic reaction can be predicted.
Learning Materials
Features
Discover
Chapter 30: Q28E (page 1033)
Predict the product of the following pericyclic reaction. Is this [5,5] shift a suprafacial or an antarafacial process?
The product of the following pericyclic reaction can be predicted.
All the tools & learning materials you need for study success - in one app.
Get started for free
What product would you expect to obtain from the photochemical cyclizationof (2E,4Z,6E)-2,4,6-octatriene? Of (2E,4Z,6Z)-2,4,6-octatriene?
Vinyl-substituted cyclopropanes undergo thermal rearrangement to yield cyclopentenes. Propose a mechanism for the reaction, and identify the pericyclic process involved.
Ring-opening of the trans-cyclobutene isomer shown takes place at much lower temperature than a similar ring-opening of the cis-cyclobutene isomer. Explain the temperature effect, and identify the stereochemistry of each reaction as either conrotatory or disrotatory.
The 1H NMR spectrum of bullvalene at 100 °C consists only of a single peak at 4.22 d. Explain.
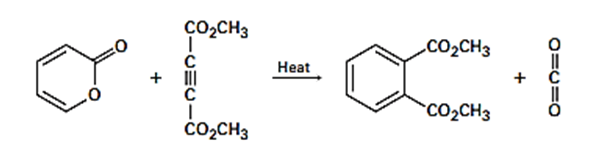
The following reaction takes place in two steps, one of which is a cycloaddition while the other is a reverse cycloaddition. Identify the two pericyclic reactions, and show how they occur.
What do you think about this solution?
We value your feedback to improve our textbook solutions.