Chapter 30: Q29E (page 1033)
Propose a pericyclic mechanism to account for the following transformation.
Short Answer
The pericyclic mechanism for the following transformation can be proposed.
Learning Materials
Features
Discover
Chapter 30: Q29E (page 1033)
Propose a pericyclic mechanism to account for the following transformation.
The pericyclic mechanism for the following transformation can be proposed.
All the tools & learning materials you need for study success - in one app.
Get started for free
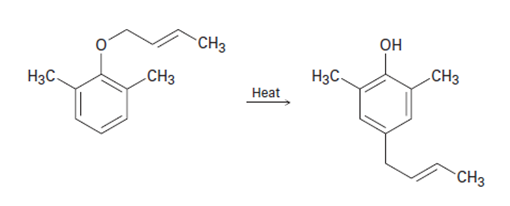
When a 2,6-disubstituted allyl phenyl ether is heated in an attempted Claisen rearrangement, migration occurs to give the p-allyl product as the result oftwo sequential pericyclic reactions. Explain.
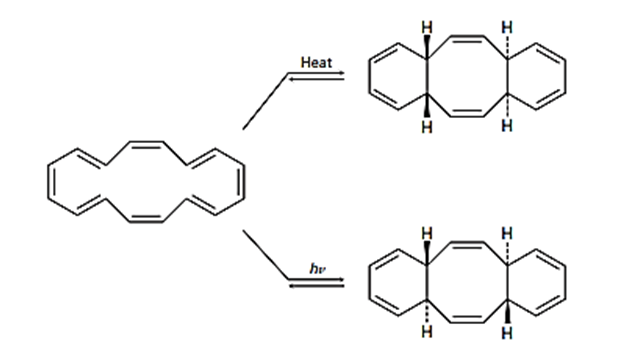
The cyclohexadecaoctaene shown isomerizes to two different isomers, depending on reaction conditions. Explain the observed results, and indicate whether each reaction is conrotatory or disrotatory.
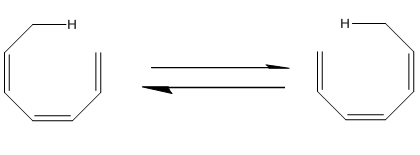
Classify the following sigmatropic reaction by order [x,y], and tell whether itwill proceed with suprafacial or antarafacial stereochemistry:
Karahanaenone, a terpenoid isolated from oil of hops, has been synthesized by the thermal reaction shown. Identify the kind of pericyclic reaction, and explain how karahanaenone is formed.
(2E,4Z,6Z,8E)-2,4,6,8-Decatetraene has been cyclized to give 7,8-dimethyl-1,3,5-cyclooctatriene. Predict the manner of ring-closure- conrotatory or disrotatory for both thermal and photochemical reactions, and predict the stereochemistry of the product in each case.
What do you think about this solution?
We value your feedback to improve our textbook solutions.