Chapter 30: Q25 E (page 1033)
Which of the following reactions is more likely to occur? Explain.
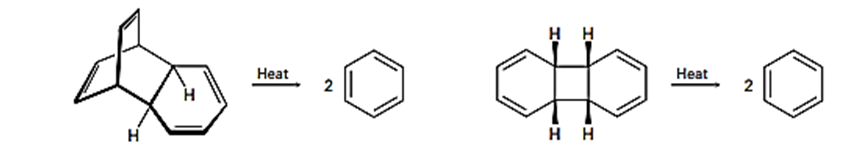
Short Answer
To identify which among the two reactions is more likely to occur can be stated.
Learning Materials
Features
Discover
Chapter 30: Q25 E (page 1033)
Which of the following reactions is more likely to occur? Explain.

To identify which among the two reactions is more likely to occur can be stated.
All the tools & learning materials you need for study success - in one app.
Get started for free
Vinyl-substituted cyclopropanes undergo thermal rearrangement to yield cyclopentenes. Propose a mechanism for the reaction, and identify the pericyclic process involved.
What stereochemistry would you expect for the product of the Diels–Alder reaction between (2E,4E)-2,4-hexadiene and ethylene? What stereochemistry would you expect if (2E,4Z)-2,4-hexadiene were used instead?
Ring-opening of the trans-cyclobutene isomer shown takes place at much lower temperature than a similar ring-opening of the cis-cyclobutene isomer. Explain the temperature effect, and identify the stereochemistry of each reaction as either conrotatory or disrotatory.
Do the following electrocyclic reactions take place in a conrotatory or
disrotatory manner? Under what conditions, thermal or photochemical, would you carry out each reaction?
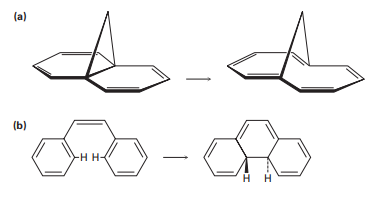
Photolysis of the cis-cyclobutene isomer in Problem 30-35 yields cis-cyclododecaen-7-yne, but photolysis of the trans isomer yields trans-cyclododecaen-7-yne. Explain these results, and identify the type and stereochemistry of the pericyclic reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.