Chapter 18: Q46E (page 594)
What product would you expect from cleavage of tetrahydrofuran
with HI?
Short Answer
The expected product from cleavage of tetrahydrofuran with HI is shown below:
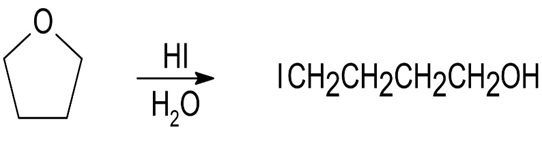
Tetrahydrofuran4-iodobutan-1-ol
Learning Materials
Features
Discover
Chapter 18: Q46E (page 594)
What product would you expect from cleavage of tetrahydrofuran
with HI?
The expected product from cleavage of tetrahydrofuran with HI is shown below:

Tetrahydrofuran4-iodobutan-1-ol
All the tools & learning materials you need for study success - in one app.
Get started for free
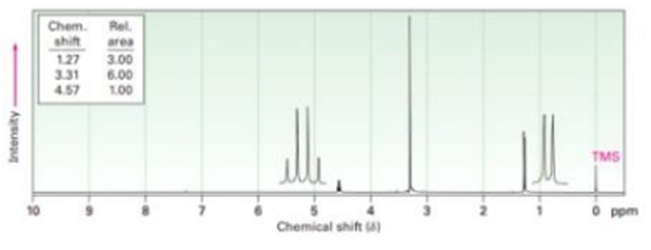
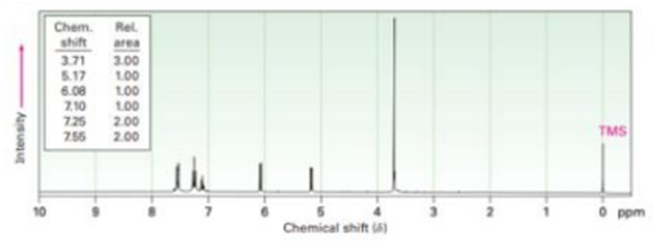
Propose structures for compounds that have the following 1H NMR spectra:
(a)
(b)
Question: tert-Butyl ethers can be prepared by the reaction of an alcohol with2-methylpropene in the presence of an acid catalyst. Propose a mechanismfor this reaction
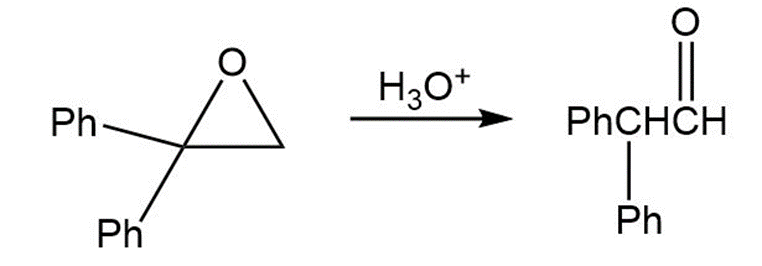
Treatment of 1,1-diphenyl-1,2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction.
Predict the product(s) and provide the mechanism for each reaction below. What do the mechanisms have in common?
a.
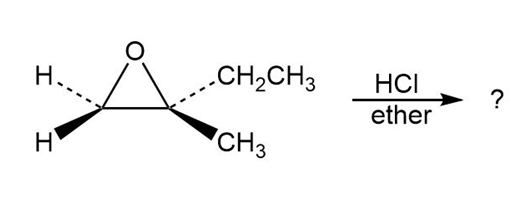
b.
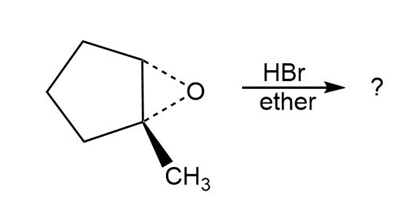
c.
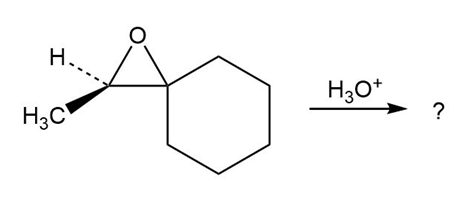
d.
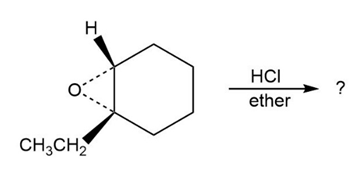
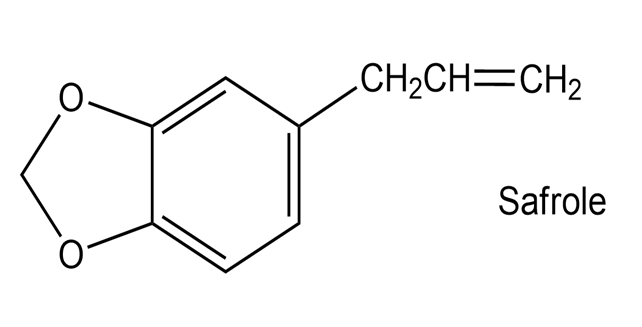
Safrole, a substance isolated from the oil of sassafras, is used as a perfumery agent. Propose a synthesis of safrole from catechol (1,2-benzenediol).
What do you think about this solution?
We value your feedback to improve our textbook solutions.