Chapter 14: Q8P (page 436)
Which of the following alkenes would you expect to be good Diels–Alder dienophiles?
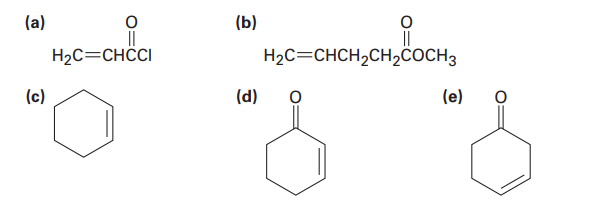
Short Answer
Compound (a) Acryloyl chloride is a good dienophile.
Compound (b) methyl pent-4-enoate is a not good dienophile.
Compound (c) cyclohexene is a not good dienophile.
Compound (d) cyclohex-2-enone is a good dienophile.
Compound (e) cyclo-3-enone is a not good dienophile.