Chapter 14: Q5P (page 430)
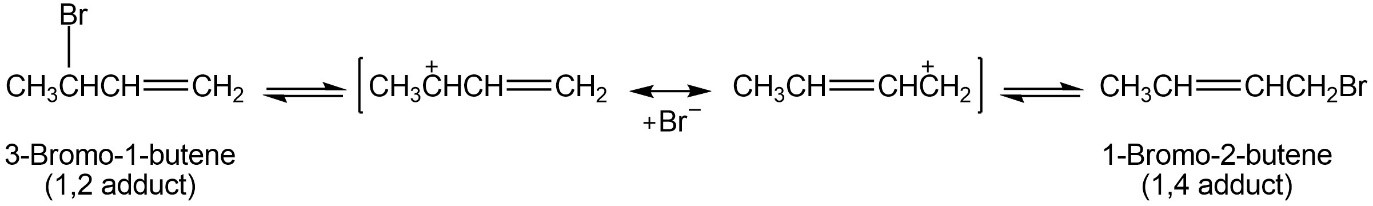
The 1,2 adduct and the 1,4 adduct formed by reaction of HBr with 1,3-butadiene are in equilibrium at 40 °C. Propose a mechanism by which the interconversion of products takes place.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q5P (page 430)
The 1,2 adduct and the 1,4 adduct formed by reaction of HBr with 1,3-butadiene are in equilibrium at 40 °C. Propose a mechanism by which the interconversion of products takes place.

All the tools & learning materials you need for study success - in one app.
Get started for free
Hydrocarbon A, C10H14 , has a UV absorption at and gives hydrocarbon B, C10H18, on catalytic hydrogenation. Ozonolysis of A, followed by zinc/acetic acid treatment, yields the following diketo dialdehyde:
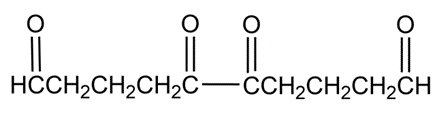
b)Hydrocarbon A reacts with maleic anhydride to yield a Diels-Alder adduct. Which of your structures for A is correct?
The following model is that of an allylic carbocation intermediateformed by protonation of a conjugated diene with HBr. Show the structureof the diene and the structures of the final reaction products.
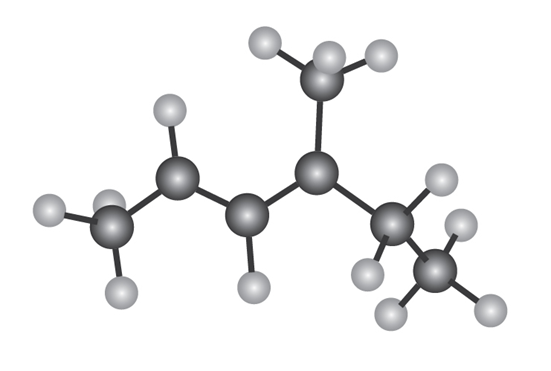
Why do you suppose 1,4 adducts of 1,3-butadiene are generally more stable than 1,2 adducts?
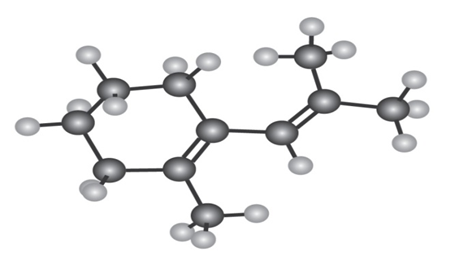
The following Diene does not undergo Diels–Alder reactions. Explain.
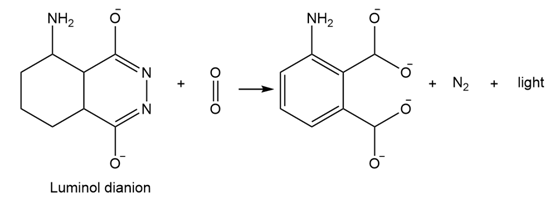
Luminol, which is used by forensic scientists to find blood, fluoresces as a result of Diels-Alder like process. The dianion of luminal reacts with to form an unstable peroxide intermediate that then loses nitrogen to form a dicarboxylate and emit light. The process is similar to that in 14-21 and 14-22. Propose a mechanism for this process.
What do you think about this solution?
We value your feedback to improve our textbook solutions.