Chapter 14: Q14-19E (page 447)
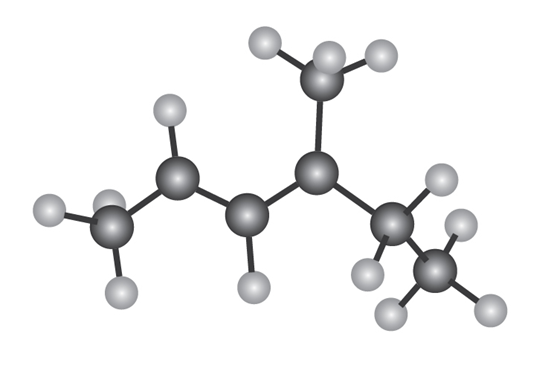
The following model is that of an allylic carbocation intermediateformed by protonation of a conjugated diene with HBr. Show the structureof the diene and the structures of the final reaction products.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q14-19E (page 447)
The following model is that of an allylic carbocation intermediateformed by protonation of a conjugated diene with HBr. Show the structureof the diene and the structures of the final reaction products.


All the tools & learning materials you need for study success - in one app.
Get started for free
Allene, has a heat of hydrogenation of
. Rank a conjugated diene, a nonconjugated diene, and an
allene in order of stability.
Why do you suppose 1,4 adducts of 1,3-butadiene are generally more stable than 1,2 adducts?
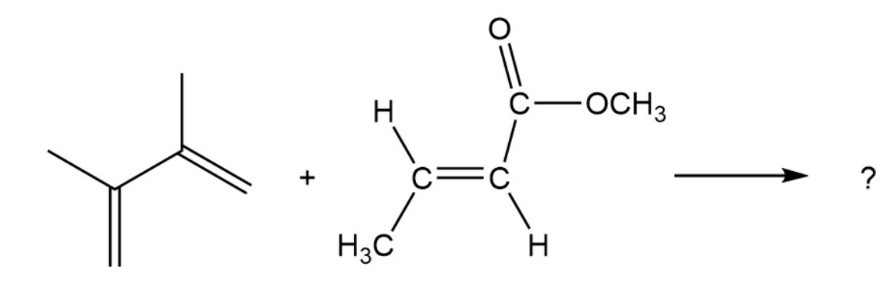
Predict the product of the following Diels–Alder reaction:
1,3-Cyclopentadiene polymerizes slowly at room temperature to yield
a polymer that has no double bonds except on the ends. On heating, the
polymer breaks down to regenerate 1,3-cyclopentadiene. Propose a structure for the product.
Dimethyl butynedioate also undergoes a Diel-Alder reaction with (2E, 4Z)-2,4-hexadiene, but the stereochemistry of the product is different from that of the (2E,4E) isomer (Problem 14-62). Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.