Chapter 14: Q1P (page 425)
Allene, has a heat of hydrogenation of
. Rank a conjugated diene, a nonconjugated diene, and an
allene in order of stability.
Short Answer
The stability order of a given alkene is as follows:
Learning Materials
Features
Discover
Chapter 14: Q1P (page 425)
Allene, has a heat of hydrogenation of
. Rank a conjugated diene, a nonconjugated diene, and an
allene in order of stability.
The stability order of a given alkene is as follows:
All the tools & learning materials you need for study success - in one app.
Get started for free
Tires whose sidewalls are made of natural rubber tend to crack and
weather rapidly in areas around cities where high levels of ozone and
other industrial pollutants are found. Explain.
Question: In the light of your answer to problem to propose mechanism for the reactions below.
Give the structures of both 1,2 and 1,4 adducts resulting from reaction of 1 equivalent of HBr with the following compound:
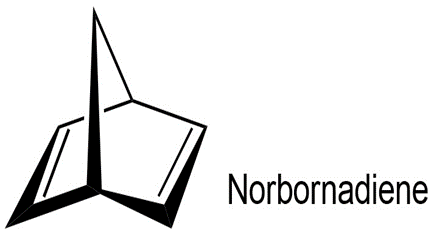
Aldrin, a chlorinated insecticide now banned from use in the United
States, can be made by Diels–Alder reaction of hexachloro-1,3-cyclopentadienewith norbornadiene. What is the structure of aldrin?
Question: 1,3,5-Hexatriene has =258 nm. In light of your answer to Problem
14-48, approximately where would you expect 2,3-dimethyl-1,3,5-hexatriene to absorb?
What do you think about this solution?
We value your feedback to improve our textbook solutions.