Chapter 14: Q20E (page 447)
Predict the major product (s) from the addition of 1 equivalent of HX and show the mechanism for each reaction below.
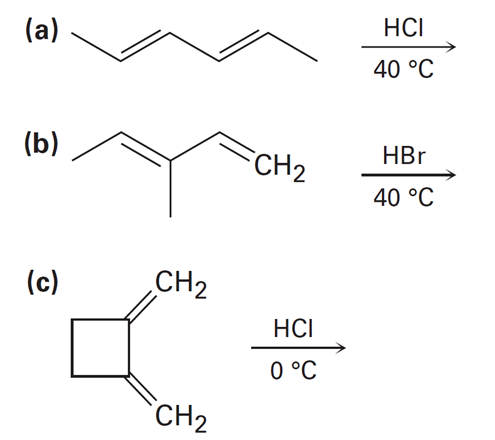
Short Answer
Step-by-Step-Solution
Learning Materials
Features
Discover
Chapter 14: Q20E (page 447)
Predict the major product (s) from the addition of 1 equivalent of HX and show the mechanism for each reaction below.

Step-by-Step-Solution
All the tools & learning materials you need for study success - in one app.
Get started for free
Would you expect allene, , to show a UV absorption in
the 200 to 400 nm range? Explain.
Draw and name the six possible diene isomers of formula. Which of the six are conjugated dienes?
Electrophilic addition of to isoprene (2-methyl-1,3-butadiene) yields the following product mixture:
Of the 1,2-addition products, explain why 3,4-dibromo-3-methyl-1-butene (21%) predominates over 3,4-dibromo-2-methyl-1-butene (3%).
Would you expect a conjugated diyne such as 1,3-butadiyne to undergo Diels-Alder reaction with a dienophile? Explain.
Which of the following alkenes would you expect to be good Diels–Alder dienophiles?
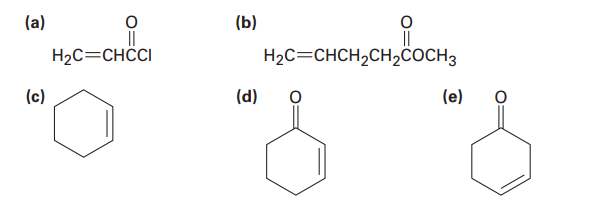
What do you think about this solution?
We value your feedback to improve our textbook solutions.