Chapter 14: Q4P (page 427)
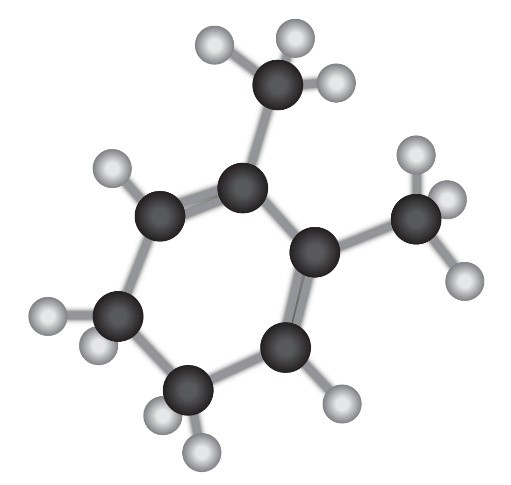
Give the structures of both 1,2 and 1,4 adducts resulting from reaction of 1 equivalent of HBr with the following compound:
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q4P (page 427)
Give the structures of both 1,2 and 1,4 adducts resulting from reaction of 1 equivalent of HBr with the following compound:


All the tools & learning materials you need for study success - in one app.
Get started for free
Would you expect a conjugated diyne such as 1,3-butadiyne to undergo Diels-Alder reaction with a dienophile? Explain.
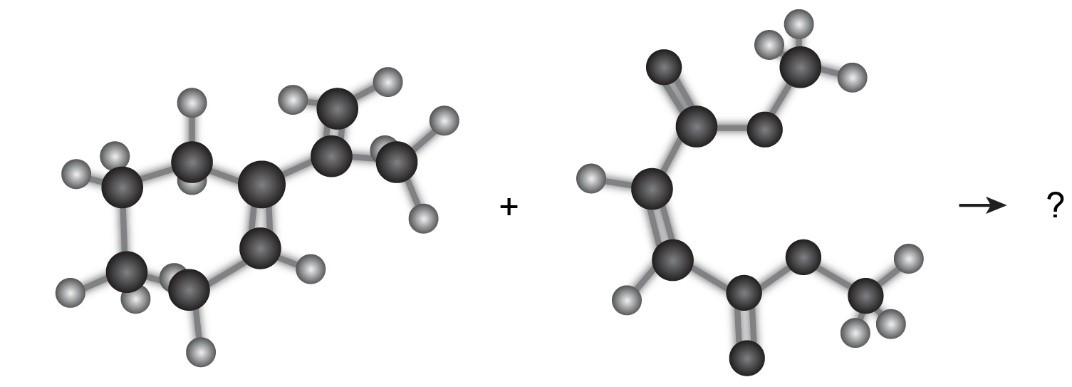
Predict the product of the following Diels–Alder reaction:
1,3-Cyclopentadiene polymerizes slowly at room temperature to yield
a polymer that has no double bonds except on the ends. On heating, the
polymer breaks down to regenerate 1,3-cyclopentadiene. Propose a structure for the product.
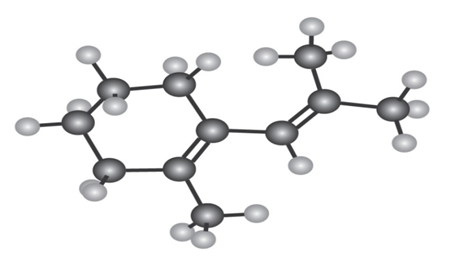
The following Diene does not undergo Diels–Alder reactions. Explain.
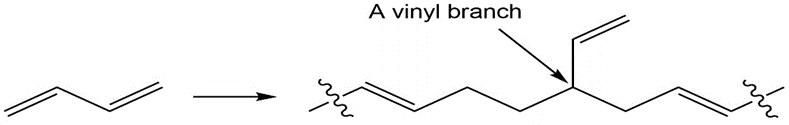
Diene polymers contain occasional vinyl branches along the chain.
How do you think these branches might arise?
What do you think about this solution?
We value your feedback to improve our textbook solutions.