Chapter 14: Q2P (page 427)
Give the structures of both 1,2 and 1,4 adducts resulting from reaction of 1 equivalent of HCl with 1,3-pentadiene.
Short Answer

Learning Materials
Features
Discover
Chapter 14: Q2P (page 427)
Give the structures of both 1,2 and 1,4 adducts resulting from reaction of 1 equivalent of HCl with 1,3-pentadiene.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: In the light of your answer to problem to propose mechanism for the reactions below.
Allene, has a heat of hydrogenation of
. Rank a conjugated diene, a nonconjugated diene, and an
allene in order of stability.
Would you expect allene, , to show a UV absorption in
the 200 to 400 nm range? Explain.
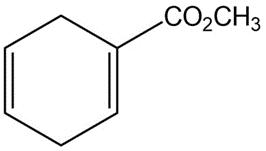
How could you use Diels-Alder reactions to prepare the following products ? Show the starting diene and dienophile in each case.
a)
The following ultraviolet absorption maxima have been measured:
1,3-Butadiene 217 nm
2-Methyl-1,3-butadiene 220 nm
1,3-Pentadiene 223 nm
2,3-Dimethyl-1,3-butadiene 226 nm
2,4-Hexadiene227 nm
2,4-Dimethyl-1,3-pentadiene 232 nm
2,5-Dimethyl-2,4-hexadiene 240 nm
What conclusion can you draw about the effect of alkyl substitution on
UV absorption maxima? Approximately what effect does each added
alkyl group have?
What do you think about this solution?
We value your feedback to improve our textbook solutions.