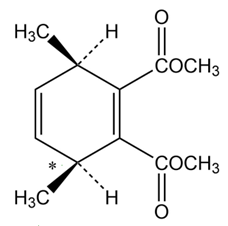
Chapter 14: Q 14-14-63 E (page 447)
Dimethyl butynedioate also undergoes a Diel-Alder reaction with (2E, 4Z)-2,4-hexadiene, but the stereochemistry of the product is different from that of the (2E,4E) isomer (Problem 14-62). Explain.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q 14-14-63 E (page 447)
Dimethyl butynedioate also undergoes a Diel-Alder reaction with (2E, 4Z)-2,4-hexadiene, but the stereochemistry of the product is different from that of the (2E,4E) isomer (Problem 14-62). Explain.

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the resonance forms that result when the dienes below are protonated. If the resonance forms differ in energy, identify the most stableone.
a)
b)
c)
Electrophilic addition of to isoprene (2-methyl-1,3-butadiene) yields the following product mixture:
Of the 1,2-addition products, explain why 3,4-dibromo-3-methyl-1-butene (21%) predominates over 3,4-dibromo-2-methyl-1-butene (3%).
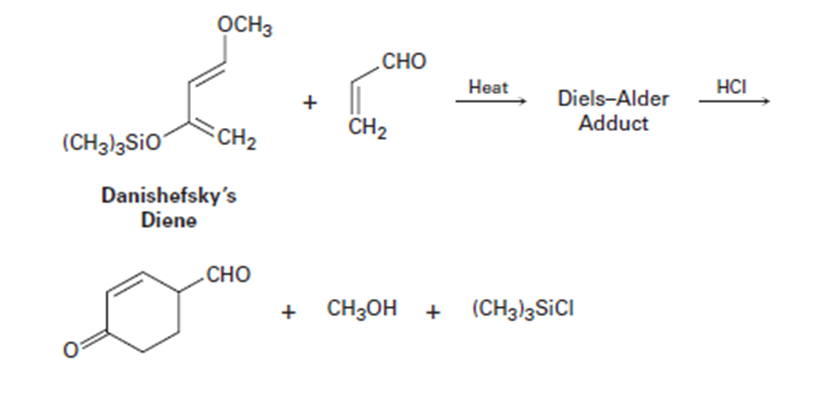
An extremely useful diene in the synthesis of many natural products is
known as Danishefsky’s diene. This compound is useful because after
the Diels–Alder reaction it can be converted into a product that could
not be accessed by a typical Diels–Alder reaction. Show the Diels–Alder
adduct and propose a mechanism that accounts for the final products.
Predict the major product (s) from the addition of 1 equivalent of HX and show the mechanism for each reaction below.
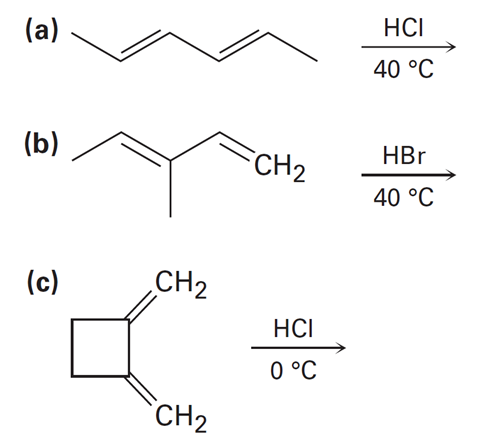
Why do you suppose 1,4 adducts of 1,3-butadiene are generally more stable than 1,2 adducts?
What do you think about this solution?
We value your feedback to improve our textbook solutions.