Chapter 14: Q14-14P (page 441)
If pure vitamin A has =5 325 (= 50,100), what is the vitamin A concentration in a sample whose absorbance at 325 nm is A = 0.735 in a cell with a path length of 1.00 cm?
Short Answer
The concentration of vitamin A is.
Learning Materials
Features
Discover
Chapter 14: Q14-14P (page 441)
If pure vitamin A has =5 325 (= 50,100), what is the vitamin A concentration in a sample whose absorbance at 325 nm is A = 0.735 in a cell with a path length of 1.00 cm?
The concentration of vitamin A is.
All the tools & learning materials you need for study success - in one app.
Get started for free
1,3-Cyclopentadiene polymerizes slowly at room temperature to yield
a polymer that has no double bonds except on the ends. On heating, the
polymer breaks down to regenerate 1,3-cyclopentadiene. Propose a structure for the product.
Give IUPAC names for the following compounds:
Question: In the light of your answer to problem to propose mechanism for the reactions below.
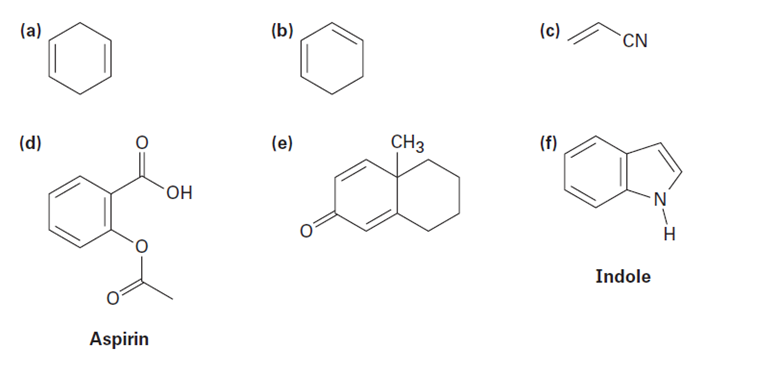
Which of the following compounds would you expect to show ultraviolet absorptions in the 200 to 400 nm range?
Predict the products of the following Diels-Alder reaction:
(b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.