Chapter 14: Q14-16E (page 447)
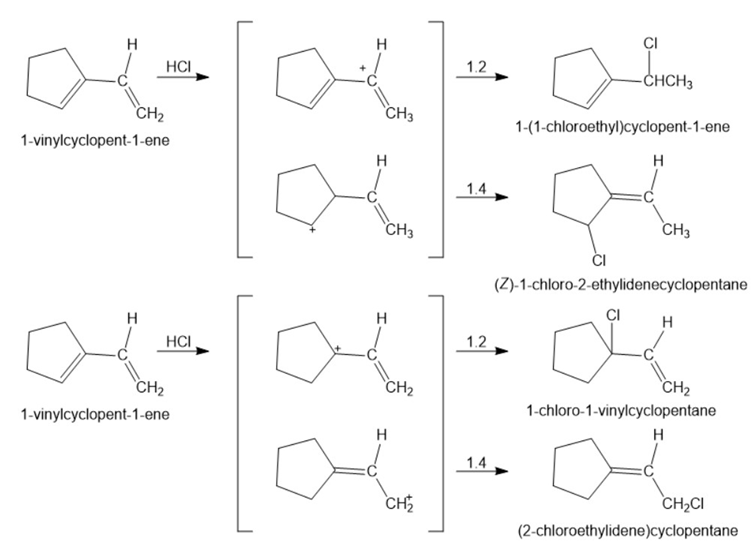
Show the structures of all possible adducts of the following diene with1 equivalent of HCl:
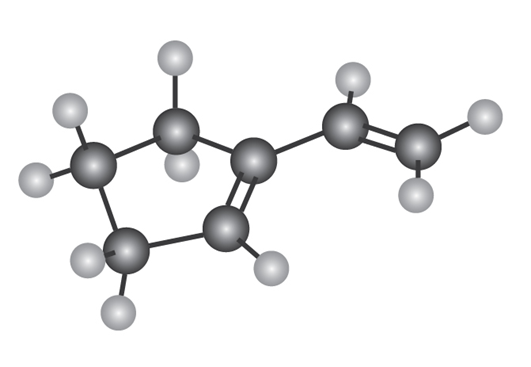
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q14-16E (page 447)
Show the structures of all possible adducts of the following diene with1 equivalent of HCl:


All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the resonance forms that result when the dienes below are protonated. If the resonance forms differ in energy, identify the most stableone.
a)
b)
c)
Show the structure, including stereochemistry, of the product from the following Diels-Alder reaction:
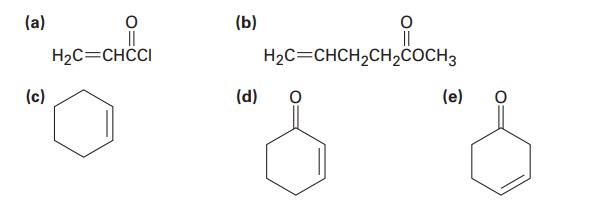
Which of the following alkenes would you expect to be good Diels–Alder dienophiles?
Treatment of 3,4-dibromohexane with strong base leads to loss of2 equivalents of HBr and formation of a product with formula C6H10.Three products are possible. Name each of the three, and tell how youwould use 1H and 13C NMR spectroscopy to help identify them. Howwould you use UV spectroscopy?
1,3-Cyclopentadiene polymerizes slowly at room temperature to yield
a polymer that has no double bonds except on the ends. On heating, the
polymer breaks down to regenerate 1,3-cyclopentadiene. Propose a structure for the product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.