Chapter 14: Q11P (page 438)
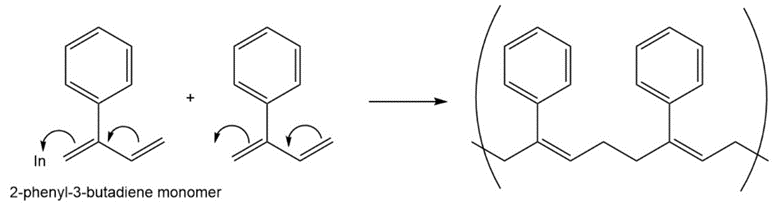
Draw a segment of the polymer that might be prepared from 2-phenyl-1,3-butadiene.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q11P (page 438)
Draw a segment of the polymer that might be prepared from 2-phenyl-1,3-butadiene.

All the tools & learning materials you need for study success - in one app.
Get started for free
How could you use Diels-Alder reactions to prepare the following products ? Show the starting diene and dienophile in each case.
a)
Tires whose sidewalls are made of natural rubber tend to crack and
weather rapidly in areas around cities where high levels of ozone and
other industrial pollutants are found. Explain.
Draw the resonance forms that result when the dienes below are protonated. If the resonance forms differ in energy, identify the most stableone.
a)
b)
c)
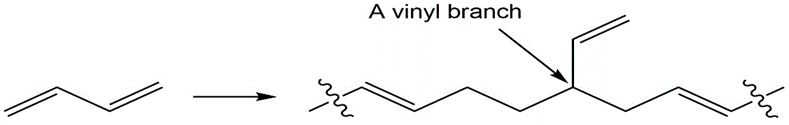
Diene polymers contain occasional vinyl branches along the chain.
How do you think these branches might arise?
Give the structures of both 1,2 and 1,4 adducts resulting from reaction of 1 equivalent of HCl with 1,3-pentadiene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.