Chapter 14: Q14-12 P (page 438)
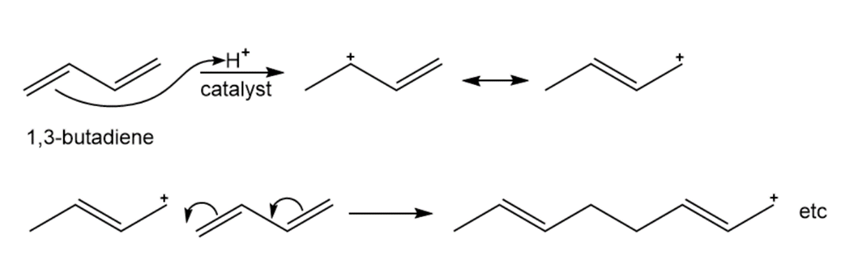
Show the mechanism of the acid-catalyzed polymerization of 1,3-butadiene.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q14-12 P (page 438)
Show the mechanism of the acid-catalyzed polymerization of 1,3-butadiene.

All the tools & learning materials you need for study success - in one app.
Get started for free
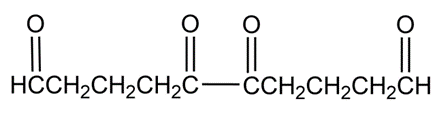
Hydrocarbon A, C10H14 , has a UV absorption at and gives hydrocarbon B, C10H18, on catalytic hydrogenation. Ozonolysis of A, followed by zinc/acetic acid treatment, yields the following diketo dialdehyde:
b)Hydrocarbon A reacts with maleic anhydride to yield a Diels-Alder adduct. Which of your structures for A is correct?
An extremely useful diene in the synthesis of many natural products is
known as Danishefsky’s diene. This compound is useful because after
the Diels–Alder reaction it can be converted into a product that could
not be accessed by a typical Diels–Alder reaction. Show the Diels–Alder
adduct and propose a mechanism that accounts for the final products.

Draw and name the six possible diene isomers of formula. Which of the six are conjugated dienes?
Predict the products of the following Diels-Alder reaction:
Would you expect a conjugated diyne such as 1,3-butadiyne to undergo Diels-Alder reaction with a dienophile? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.