Chapter 16: Q64E (page 524)
How would you synthesize the following substances starting frombenzene?
(a)
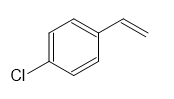
(b)
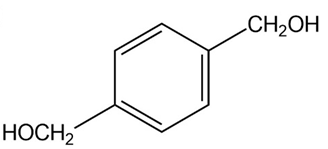
(c)
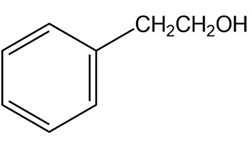
Short Answer
(a)
 Synthesis of compound (a)
Synthesis of compound (a)
(b)
 Synthesis of compound (b)
Synthesis of compound (b)
(c)
 Synthesis of compound (c)
Synthesis of compound (c)
Learning Materials
Features
Discover
Chapter 16: Q64E (page 524)
How would you synthesize the following substances starting frombenzene?
(a)

(b)

(c)

(a)
 Synthesis of compound (a)
Synthesis of compound (a)
(b)
 Synthesis of compound (b)
Synthesis of compound (b)
(c)
 Synthesis of compound (c)
Synthesis of compound (c)
All the tools & learning materials you need for study success - in one app.
Get started for free
At what position, and on what ring, would you expect the followingsubstances to undergo electrophilic substitution?
(a)
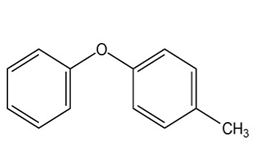
(b)
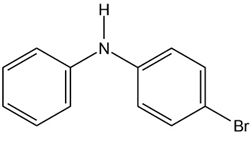
(c)
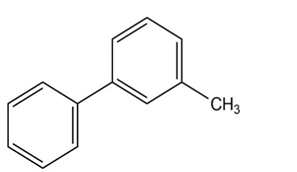
(d)
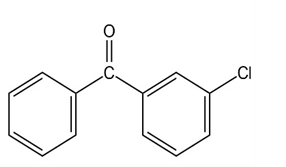
What product(s) would you expect to obtain from the following
reactions?
(a)
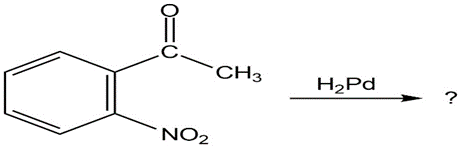
(b)
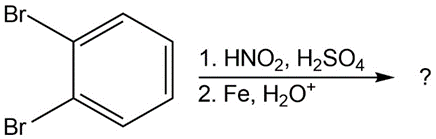
(c)
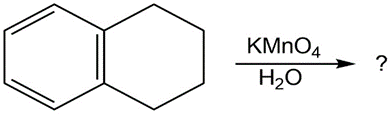
(d)
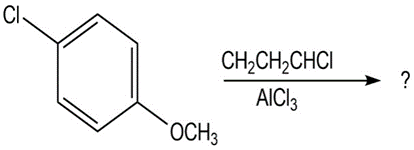
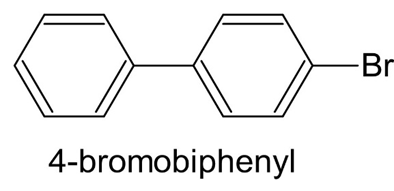
At what position and on what ring do you expect nitration of 4-bromobiphenyl to occur? Explain, using resonance structures of the potentialintermediates.
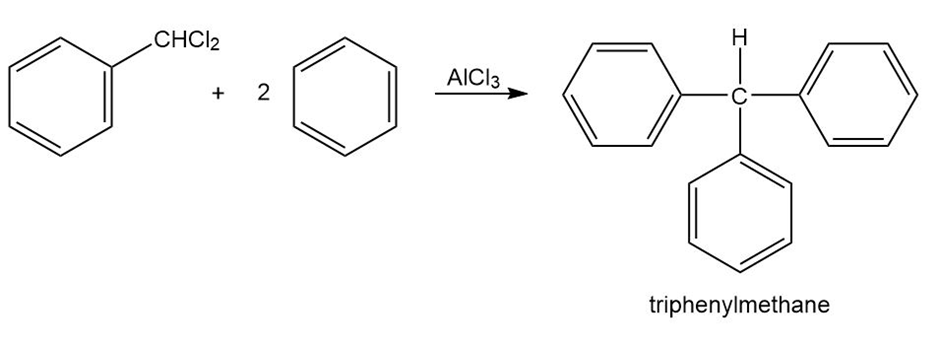
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of . Propose a mechanism for the reaction.
Question: Would you expect p-methyl phenol to be more acidic or less acidic than unsubstituted phenol? Explain. (See Problem 16-75.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.