Chapter 16: Q16-21P (page 514)
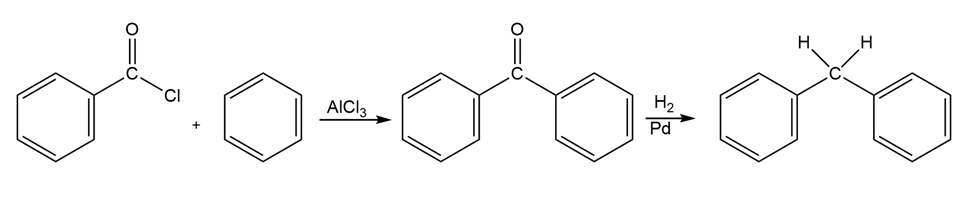
How would you prepare diphenylmethane, , from benzene and an acid chloride?
Short Answer
Learning Materials
Features
Discover
Chapter 16: Q16-21P (page 514)
How would you prepare diphenylmethane, , from benzene and an acid chloride?

All the tools & learning materials you need for study success - in one app.
Get started for free
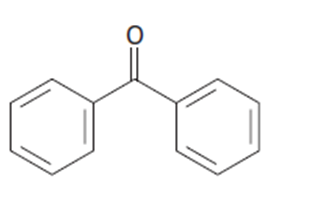
Identify the carboxylic acid chloride that might be used in a Friedel–Crafts acylation reaction to prepare each of the following acylbenzenes:
a.
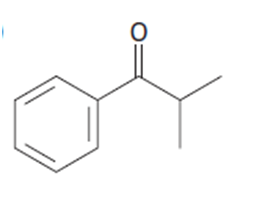
b.
Starting with benzene as your only source of aromatic compounds,
how would you synthesize the following substances? Assume that you
can separate ortho and para isomers if necessary.
(a)p-Chloroacetophenone (b)m-Bromonitrobenzene
(c)o-Bromobenzenesulfonic acid (d)m-Chlorobenzenesulfonic acid
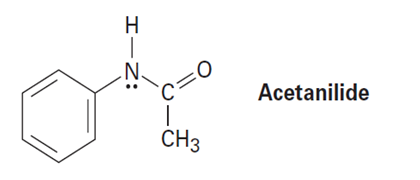
Acetanilide is less reactive than aniline toward electrophilic substitution. Explain
Rank the compounds in each of the following groups in order of their reactivity
to electrophilic substitution:
(a) Nitrobenzene, phenol, toluene, benzene
(b) Phenol, benzene, chlorobenzene, benzoic acid
(c) Benzene, bromobenzene, benzaldehyde, aniline
Electrophilic substitution on 3-phenylpropanenitrile occurs at theortho and para positions, but reaction with 3-phenylpropenenitrileoccurs at the meta position. Explain, using resonance structures of theintermediates.
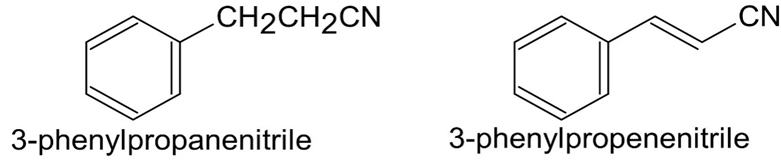
What do you think about this solution?
We value your feedback to improve our textbook solutions.