Chapter 16: Q7 P (page 493)
Identify the carboxylic acid chloride that might be used in a Friedel–Crafts acylation reaction to prepare each of the following acylbenzenes:
a.
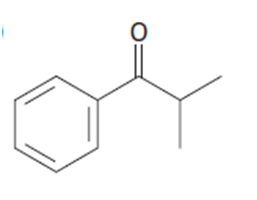
b.
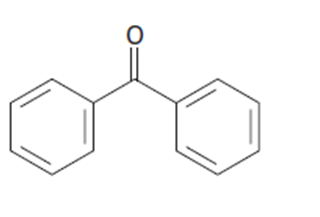
Short Answer
a.
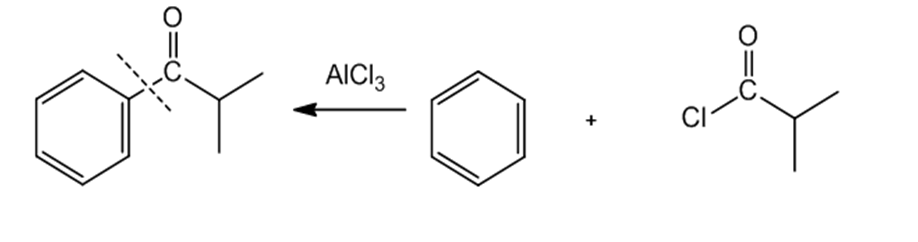
b.
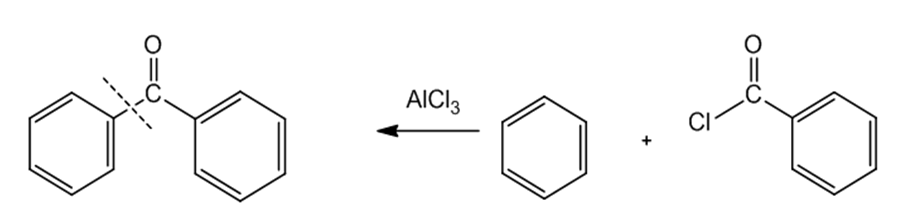
Learning Materials
Features
Discover
Chapter 16: Q7 P (page 493)
Identify the carboxylic acid chloride that might be used in a Friedel–Crafts acylation reaction to prepare each of the following acylbenzenes:
a.

b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
How many products might be formed on chlorination of o-xylene (o-dimethylbenzene), m-xylene, and p-xylene?
Would you expect the Friedel–Crafts reaction of benzene with (R)-2-chlorobutane to yield optically active or racemic product? Explain.
How would you synthesize the following substances starting from benzene or phenol? Assume that ortho- and para-substitution products can
be separated.
(a)o-Bromobenzoic acid (b)p-Methoxytoluene
(c)2,4,6-Trinitrobenzoic acid (d)m-Bromoaniline
Refer to Table 6-3 on page 170 for a quantitative idea of the stability of a benzyl radical. How much more stable (in kJ/mol) is the benzyl radical than a primary alkyl radical? How does a benzyl radical compare in stability to an allylradical?
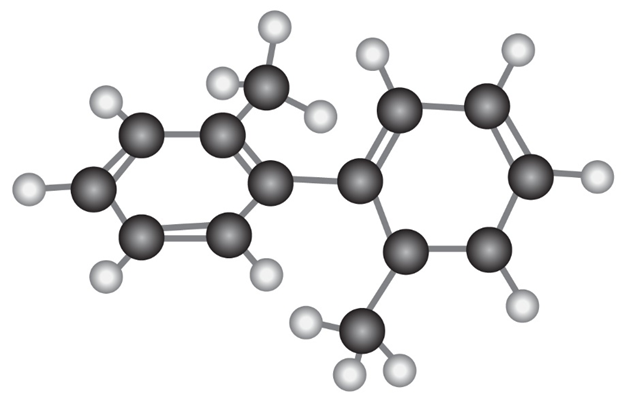
The following molecular model of a dimethyl-substituted biphenyl represents the lowest-energy conformation of the molecule. Why are the two benzene rings tilted at a 63° angle to each other rather than being in the same plane so that their p orbitals overlap? Why doesn’t complete rotation around the single bond joining the two rings occur?
What do you think about this solution?
We value your feedback to improve our textbook solutions.