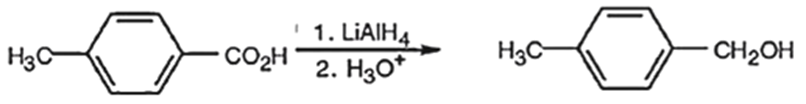
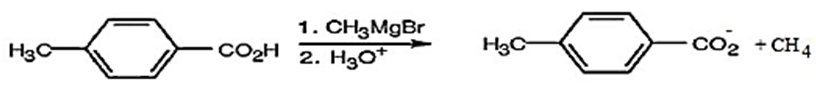(a) When p-methyl benzoic acid is treated with the carboxyl group is reduced to a primary alcohol group. The product obtained is p-methyl-benzyl alcohol.
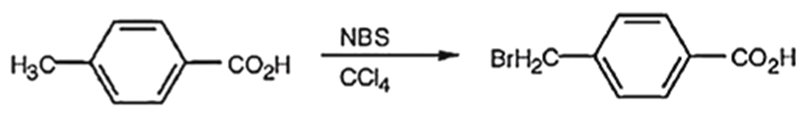
(b) When p-methyl benzoic acid is treated with N-bromosuccinimide in , a bromine atom gets substituted in the benzyl carbon. The product obtained is p-bromomethylbenzoic acid.
(c) When p-methyl benzoic acid is treated with the methyl anion in the Grignard reagent picks up a proton from the acid to obtain a product called methane.
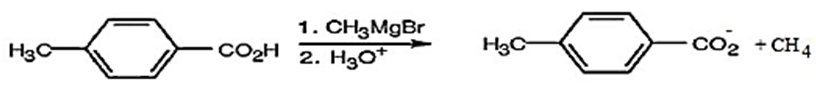
(d) When p-methyl benzoic acid is treated with in the presence of the methyl group in the p-position is oxidized to acid. The product obtained is benzene-1,4-dicarboxylic acid.