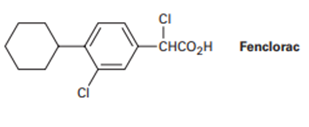
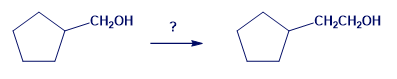Chapter 20: Q43P (page 678)
How could you convert butanoic acid into the following compounds?
Write each step showing the reagents needed.
(a) 1-Butanol
(b) 1-Bromobutane
(c) Pentanoic acid
(d) 1-Butene
(e) Octane
Short Answer
The conversion of butanoic acid into the following compounds can be explained.