Chapter 21: Q79E (page 726)
Propose a structure for the compound with the formulaC19H9NO2 and the following IR and NMR spectra
Short Answer
The Structure should be
Learning Materials
Features
Discover
Chapter 21: Q79E (page 726)
Propose a structure for the compound with the formulaC19H9NO2 and the following IR and NMR spectra
The Structure should be
All the tools & learning materials you need for study success - in one app.
Get started for free
21-73 N,N-Diethyl-m-toluamide (DEET) is the active ingredient in manyinsect repellent preparations. How might you synthesize this substancefrom m-bromotoluene?
Predict the products of the following nucleophilic acyl substitution reactions:
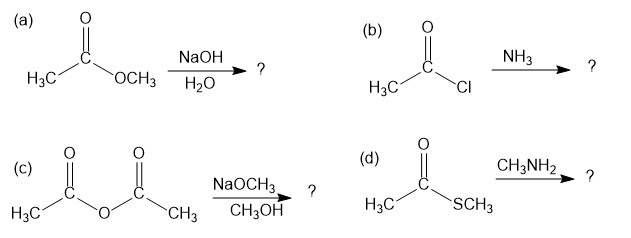
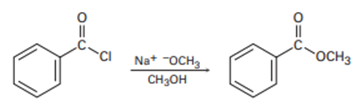
Show the mechanism of the following nucleophilic acyl substitution reaction, using curved arrows to indicate the electron flow in each step:
Draw structures corresponding to the following names:
(a) Phenyl benzoate
(b) N-Ethyl-N-methylbutanamide
(c) 2,4-Dimethylpentanoyl chloride
(d) Methyl 1-methylcyclohexanecarboxylate
(e) Ethyl 3-oxopentanoate
(f) Methyl p-bromobenzenethioate
(g) Formic propanoic anhydride
(h) cis-2-Methylcyclopentanecarbonyl bromide
21-75 One frequently used method for preparing methyl esters is by reactionof carboxylic acids with diazomethane,
The reaction occurs in two steps: (1) protonation of diazomethane bythe carboxylic acid to yield methyldiazonium ion,, plus a carboxylate ion; and (2) reaction of the carboxylate ion with .
(a) Draw two resonance structures of diazomethane, and account forstep 1.
(b) What kind of reaction occurs in step 2?
What do you think about this solution?
We value your feedback to improve our textbook solutions.