Chapter 21: Q78E (page 726)
Assign structures to compounds with the following 1H NMR spectra:
(a)C5H9ClO2
IR:1735 cm-1
Short Answer
Thestructure of the compound using 1H NMR spectra can be drawn.
Learning Materials
Features
Discover
Chapter 21: Q78E (page 726)
Assign structures to compounds with the following 1H NMR spectra:
(a)C5H9ClO2
IR:1735 cm-1
Thestructure of the compound using 1H NMR spectra can be drawn.
All the tools & learning materials you need for study success - in one app.
Get started for free
Explain the observation that attempted Fischer esterification of 2,4,6-trimethyl benzoic acid with methanol and HCL is unsuccessful. No ester is obtained, and the acid is recovered unchanged. What alternative method of esterification might be successful?
21-49 Answer Problem 21-48 for reaction of the listed reagents with methylpropanoate.
Bacteria typically develop a resistance to penicillins and other β-lactam antibiotics (see Something Extra in this chapter) due to bacterial synthesis of β -lactamase enzymes. Tazobactam, however, is able to inhibit the activity of the β -lactamase by trapping it, thereby preventing a resistance from developing.
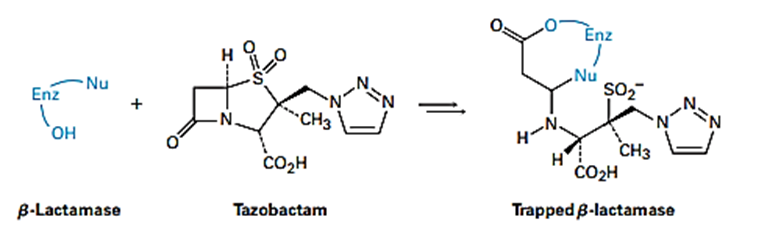
(a) The first step in trapping is the reaction of a hydroxyl group on the β -lactamase to open the β -lactam ring of tazobactam. Show the mechanism.
(b) The second step is opening the sulfur-containing ring in tazobactam to give an acyclic imine intermediate. Show the mechanism.
(c) Cyclization of the imine intermediate gives the trapped β-lactamase product. Show the mechanism.
21-69 We said in Section 21-6 that mechanistic studies on ester hydrolysishave been carried out using ethyl propanoate labeled with in theether-like oxygen. Assumethat labeled acetic acid is your onlysource of isotopic oxygen, and then propose a synthesis of the labeledethyl propanoate.
Write the mechanism of the reaction just shown between 3,4,5-trimethoxybenzoyl chloride and morpholine to form trimetozine. Use curved arrows to show the electron flow in each step.
What do you think about this solution?
We value your feedback to improve our textbook solutions.