Chapter 15: Q15-1P (page 455)
Tell whether the following compounds are ortho-, meta-, or para-disubstituted:
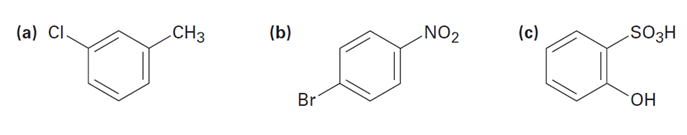
Short Answer
The name of the compounds are stated below
- meta-chloro toluene
- para-nitro chloro benzene
- ortho sulphonyl phenol
Learning Materials
Features
Discover
Chapter 15: Q15-1P (page 455)
Tell whether the following compounds are ortho-, meta-, or para-disubstituted:

The name of the compounds are stated below
All the tools & learning materials you need for study success - in one app.
Get started for free
Thiamin, or vitamin B1, contains a positively charged five-membered nitrogen–sulfur heterocycle called a thiazolium ring. Explain why the thiazolium ring is aromatic.

Propose a structure for a molecule that has the following NMR spectrum and has IR absorptions at 700, 740, and :
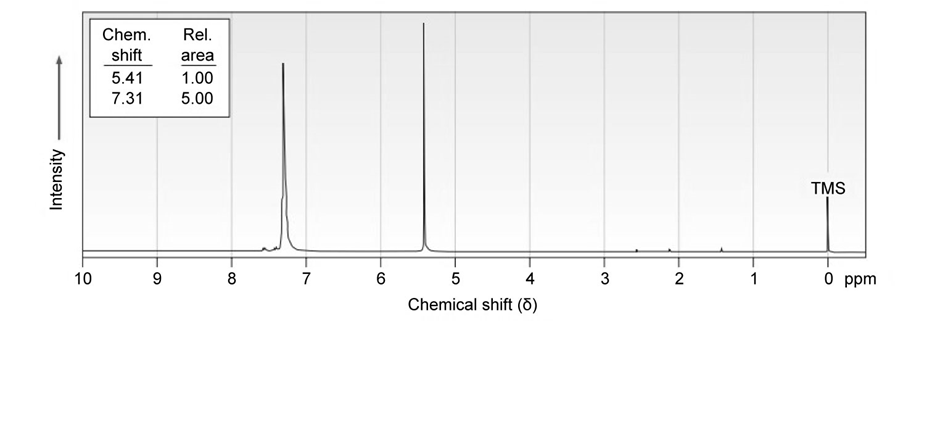
Azulene, an isomer of naphthalene, has a remarkably large dipole moment for a hydrocarbon (5 1.0 D). Explain, using resonance structures.
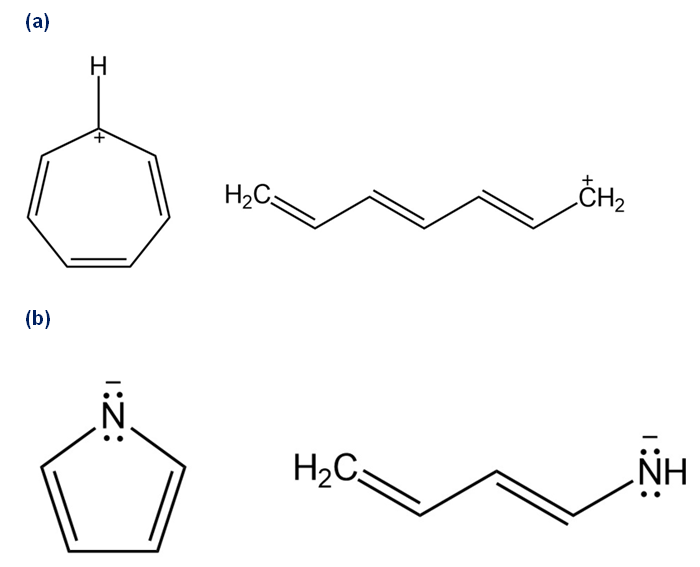
Draw all of the resonance forms for each. What patterns emerge?
The compound below is the product initially formed in a Claisen rearrangement (Section 18-4). This product is not isolated but tautomerizes to its enol form. Give the structure of the enol and provide an explanation as to why the enol tautomer is favored.

What do you think about this solution?
We value your feedback to improve our textbook solutions.